13.3
Impact Factor
Theranostics 2019; 9(18):5386-5395. doi:10.7150/thno.35032 This issue Cite
Research Paper
RNA-silencing nanoprobes for effective activation and dynamic imaging of neural stem cell differentiation
1. Engineering Research Center of Molecular- and Neuro-imaging of ministry of education, School of Life Science and Technology, Xidian University, Xi'an, Shaanxi, 710126 China.
2. Key Laboratory of Molecular Imaging, Institute of Automation, Chinese Academy of Sciences, Beijing, 100190, China.
3. Beijing Advanced Innovation Center for Big Data-Based Precision Medicine, School of Medicine, Beihang University, Beijing, 100191, China
4. Laboratory of Molecular Imaging and Nanomedicine, National Institute of Biomedical Imaging and Bioengineering, National Institutes of Health, Bethesda, Maryland, 20892 USA
Abstract
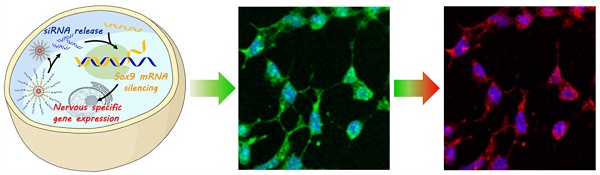
To achieve the clinical potential of neural stem cells (NSCs), it is crucial to activate NSC differentiation into neurons and simultaneously monitor the process of NSC differentiation. However, there are many challenges associated with regulating and tracking NSC differentiation.
Methods: We developed a redox-responsive multifunctional nanocomplex with a disulfide bond—cvNC—for the delivery of siRNAs to induce NSC differentiation through sequence-specific RNA interference (RNAi) and real-time imaging of sequential mRNA expression during differentiation. The stability and specificity of cvNCs were studied in vitro. Controlled release of siRNA, gene silencing efficiency, as well as real-time imaging of cvNCs on Tubb3 and Fox3 mRNAs during NSC differentiation were evaluated.
Results: The introduction of a redox-sensitive disulfide bond not only ensures the remarkable performance of cvNC, such as high stability, controlled siRNA release, and enhanced gene silencing efficiency, but also effectively stimulates NSC differentiation into neurons. More importantly, the cvNC can track NSC differentiation in real-time by monitoring the sequential expression of mRNAs.
Conclusion: Our study indicates that cvNC can serve as a robust system for exploring NSCs differentiation process as well as other biological events in living cells.
Keywords: gold nanoparticle, gene silencing, imaging, drug delivery, neural stem cell
 Global reach, higher impact
Global reach, higher impact