Impact Factor
Theranostics 2019; 9(18):5412-5423. doi:10.7150/thno.33598 This issue Cite
Research Paper
A novel anti-DR5 antibody-drug conjugate possesses a high-potential therapeutic efficacy for leukemia and solid tumors
1. Yantai Obioadc Biomedical Technology Ltd., Yantai, China
2. Obio Technology (Shanghai) Corp., Ltd., No. 908, Bldg. 19, Ziping Rd., Pudong New District, Shanghai 201321, China
3. Yantai Mabplex International Bio-Pharmaceutical Co., Ltd., Yantai, China
Abstract
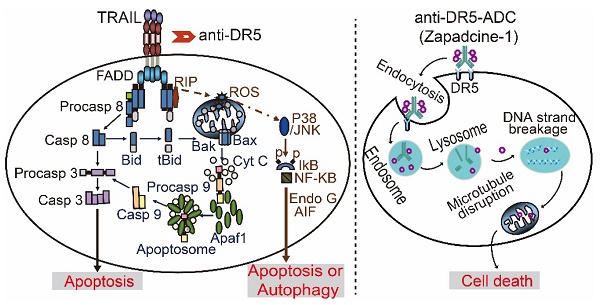
It is well known that tumor necrosis factor-related apoptosis inducing ligand receptor 1 or 2 (DR4/DR5) is specifically expressed in various tumor cells, but less or no expression in most normal cells. Many first generations of TRAIL agonists including recombinant preparations of TRAIL, agonistic antibodies against DR4/DR5 have been developed in phase I/II clinical trials for cancer therapy. However, the outcomes of clinical trials by using DR4/DR5 agonist mono-therapy were disappointed even though the safety profile was well tolerance. In the present study, we report an anti-DR5 antibody-drug conjugate (ADC, named as Zapadcine-1) possesses a higher potential for the therapy of lymphocyte leukemia and solid cancers.
Methods: Zapadcine-1 was made by a fully humanized DR5-specific monoclonal antibody (Zaptuzumab) coupled via a cleavable linker to a highly toxic inhibitor of tubulin, monomethyl auristatin D (MMAD), by using ThioBridge technology. Cytotoxicity of the ADC in various tumor cells was identified by luminescent cell viability assay and the efficacy in vivo was determined in cells derived xenografts (CDX) of Jurkat E6-1, BALL-1, Reh, and patient derived xenografts (PDX) of human acute leukemia. Preliminary safety evaluation was carried out in rat and monkey. Results: Zapadcine-1 possesses a similar binding ability to the death receptor DR5 as the naked monoclonal antibody Zaptuzumab, and can be rapidly endocytosed into the lysosome of cancer cells. Zapadcine-1 specifically kills human lymphocyte leukemia cells and solid tumor cells, but not normal cells tested. More importantly, Zapadcine-1 drastically eliminates the xenografts in both CDX and PDX models of human acute leukemia. The excellent and comparable therapeutic efficacy is also observed in lung cancer NCI-H1975 CDX mouse model. The maximum-tolerated dose (MTD) of single injected Zapadcine-1 in rat and cynomolgus monkey shows an acceptable safety profile.
Conclusion: These data demonstrate a promising anti-cancer activity, meriting further exploration of its potential as a novel cancer therapeutic agent, especially for the acute lymphocyte leukemia.
Keywords: DR5 antibody, antibody-drug conjugate, leukemia, solid tumor, therapy
 Global reach, higher impact
Global reach, higher impact