13.3
Impact Factor
Theranostics 2019; 9(19):5595-5609. doi:10.7150/thno.34669 This issue Cite
Research Paper
Ultrasound-mediated cavitation enhances the delivery of an EGFR-targeting liposomal formulation designed for chemo-radionuclide therapy
1. CRUK/MRC Oxford Institute for Radiation Oncology, Department of Oncology, University of Oxford, Oxford, OX3 7DQ, UK.
2. Institute of Biomedical Engineering, Department of Engineering Science, University of Oxford, Oxford, OX3 7DQ, UK.
*These authors contributed equally to the work.
Abstract
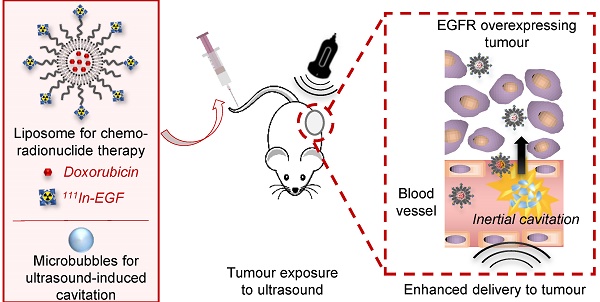
Nanomedicines allow active targeting of cancer for diagnostic and therapeutic applications through incorporation of multiple functional components. Frequently, however, clinical translation is hindered by poor intratumoural delivery and distribution. The application of physical stimuli to promote tumour uptake is a viable route to overcome this limitation. In this study, ultrasound-mediated cavitation of microbubbles was investigated as a mean of enhancing the delivery of a liposome designed for chemo-radionuclide therapy targeted to EGFR overexpressing cancer. Method: Liposomes (111In-EGF-LP-Dox) were prepared by encapsulation of doxorubicin (Dox) and surface functionalisation with Indium-111 tagged epidermal growth factor. Human breast cancer cell lines with high and low EGFR expression (MDA-MB-468 and MCF7 respectively) were used to study selectivity of liposomal uptake, subcellular localisation of drug payload, cytotoxicity and DNA damage. Liposome extravasation following ultrasound-induced cavitation of microbubbles (SonoVue®) was studied using a tissue-mimicking phantom. In vivo stability, pharmacokinetic profile and biodistribution were evaluated following intravenous administration of 111In-labelled, EGF-functionalised liposomes to mice bearing subcutaneous MDA-MB-468 xenografts. Finally, the influence of ultrasound-mediated cavitation on the delivery of liposomes into tumours was studied. Results: Liposomes were loaded efficiently with Dox, surface decorated with 111In-EGF and showed selective uptake in MDA-MB-468 cells compared to MCF7. Following binding to EGFR, Dox was released into the intracellular space and 111In-EGF shuttled to the cell nucleus. DNA damage and cell kill were higher in MDA-MB-468 than MCF7 cells. Moreover, Dox and 111In were shown to have an additive cytotoxic effect in MDA-MB-468 cells. US-mediated cavitation increased the extravasation of liposomes in an in vitro gel phantom model. In vivo, the application of ultrasound with microbubbles increased tumour uptake by 66% (p<0.05) despite poor vascularisation of MDA-MB-468 xenografts (as shown by DCE-MRI). Conclusion: 111In-EGF-LP-Dox designed for concurrent chemo-radionuclide therapy showed specificity for and cytotoxicity towards EGFR-overexpressing cancer cells. Delivery to tumours was enhanced by the use of ultrasound-mediated cavitation indicating that this approach has the potential to deliver cytotoxic levels of therapeutic radionuclide to solid tumours.
Keywords: Ultrasound-Enhanced Delivery, Radionuclide Therapy, Chemotherapy, Breast Cancer, Liposome.
 Global reach, higher impact
Global reach, higher impact