13.3
Impact Factor
Theranostics 2019; 9(19):5610-5625. doi:10.7150/thno.34755 This issue Cite
Research Paper
Dual-Targeted Gold Nanoprism for Recognition of Early Apoptosis, Dual-Model Imaging and Precise Cancer Photothermal Therapy
1. School of Life Sciences, Anhui Agricultural University, Hefei, 230036, China
2. Key Lab of Cerebral Microcirculation in Universities of Shandong, Shandong First Medical University & Shandong Academy of Medical Sciences, Taian, Shandong, 271000, China
*The authors contributed equally to this work.
Abstract
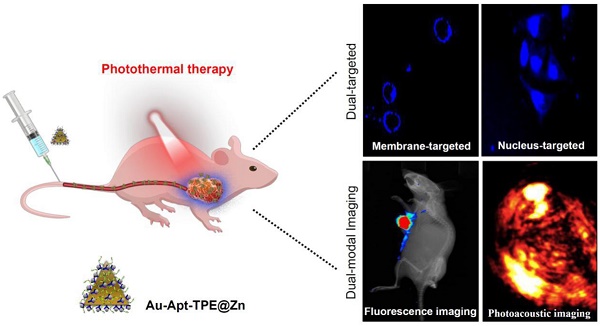
Photothermal therapy as novel strategy to convert near-infrared (NIR) light into heat for treatment cancers has attracted great attention and been widely studied. However, side effects and low efficiency remain the main challenge of precise cancer photothermal therapy.
Methods: In this study, we have successfully fabricated and characterized the dual-targeted gold nanoprisms, whereby bare gold nanoprisms (Au NPR) were conjugated to a phenanthroline derivatives-functionalized tetraphenylethene (TPE) and further stabilized with target peptide aptamers via Au-S bonds (Au-Apt-TPE). Then, the remaining nitrogen atoms of the Au-Apt-TPE could effectively chelate with Zn2+ ions (Au-Apt-TPE@Zn) for monitoring early stage apoptotic cells.
Results: The as-synthesized Au-Apt-TPE@Zn exhibited good monodispersity, size stability and consistent spectral characteristics. TPE synthesized here showed aggregation-induced emission (AIE) characteristics, and zinc conjunction (TPE@Zn) endowed Au-Apt-TPE@Zn with the cell membrane-targeted ability to selectively recognize the membranes of early stage apoptotic cells but not respond to healthy cells, which provided valuable diagnosis information on therapeutic efficacy. Au-Apt-TPE@Zn achieved specifically nuclear-targeted ability by surface decoration of AS1411 DNA aptamer. Au-Apt-TPE@Zn under NIR irradiation showed effective photothermal therapy against SGC-7901 human gastric carcinoma cells growth in vitro by inducing apoptosis through triggering reactive oxygen species (ROS) overproduction and regulating multiple signal crosstalk. In vivo studies revealed that Au-Apt-TPE@Zn under NIR irradiation showed deep penetration and dual-model imaging application (cancer-targeted fluorescence imaging and light-up photoacoustic imaging). Au-Apt-TPE@Zn under NIR irradiation also displayed strong photothermal therapy against gastric carcinoma xenograft growth in vivo by induction of apoptosis. Importantly, analysis of histopathology, hematotoxicity and immunocytotoxicity indicated that Au-Apt-TPE@Zn had less side effect and high biocompatibility.
Conclusions: Our findings validated the design of using Au nanoprism with AIE materials and dual-targeted decoration could be an effective strategy in recognition of early apoptosis, dual-model imaging and precise cancer photothermal therapy.
Keywords: Gold nanoprisms, aggregation-induced emission, apoptosis, reactive oxygen species, photothermal therapy
 Global reach, higher impact
Global reach, higher impact