13.3
Impact Factor
Theranostics 2019; 9(19):5657-5671. doi:10.7150/thno.33925 This issue Cite
Research Paper
Extracellular vesicles expressing a single-chain variable fragment of an HIV-1 specific antibody selectively target Env+ tissues
1. Center for Public Health Research, Medical School, Nanjing University, Nanjing, China,
2. State Key Laboratory of Analytical Chemistry for Life Science, Nanjing University, Nanjing, China,
3. Medical School, Jiangsu Key Laboratory of Molecular Medicine, Nanjing University, Nanjing, China
4. Model Animal Research Center, Nanjing University, China
5. Department of infectious disease, The Second Hospital of Nanjing, Nanjing University of Chinese Medicine
Abstract
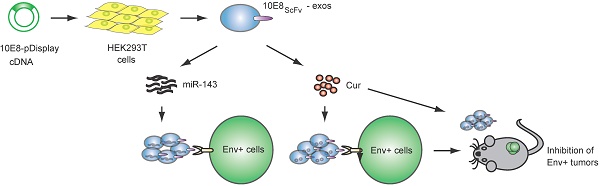
Rationale: Antiretroviral therapy can effectively suppress HIV-1 replication in the peripheral blood to an undetectable level. However, elimination of the latent virus in reservoirs remains a challenge and is a major obstacle in the treatment of HIV-1-infected patients. Exosomes exhibit huge promise as an endogenous drug delivery nanosystem for delivering drugs to solid tissues given their unique properties, including low immunogenicity, innate stability, high delivery efficiency, and most importantly the ability to penetrate solid tissues due to their lipophilic properties.
Methods: We engineered and expressed the scFv of a high affinity HIV-1-specific monoclonal antibody, 10E8, on the exosomal surface (10E8scFv-exos). Subsequently, the 10E8scFv-exos were loaded with curcumin (Cur), a chemical that kills HIV-1-infected cells, or miR-143, an apoptosis-inducing miRNA. We tested the ability of 10E8scFv-exos to deliver cargo to Env+ target cells and tissues, as well as their ability to suppress HIV-1 infection.
Results: 10E8scFv-exos efficiently targeted CHO cells expressing a trimeric gp140 on their surface (Env+ cells) in vitro, as demonstrated by confocal imaging and flow cytometry. 10E8scFv-exos loaded with Cur or miR-143 showed specific killing of Env+ cells. In addition, 10E8scFv-exos loaded with Cur or miR-143 could suppress p24 expression in an HIV-1 latency cell line ACH2 and in PBMCs from an ART-treated HIV-1-infected patient. In an NCG mouse model grafted with tumorigenic Env+ CHO cells and which had developed solid tissue tumors, intravenously injected 10E8scFv-exos targeted the Env-expressing tissues and delivered Cur to induce a strong suppression of the Env+ tumor growth with low toxicity.
Conclusion: In principle, engineered exosomes can deliver anti-HIV agents to solid tissues by specifically targeting cells expressing viral envelop proteins and inducing cell killing, suggesting that such an approach could be developed for eradicating virus-infected cells in tissue reservoirs.
Keywords: HIV-targeted delivery system, exosome, extracellular vesicles, chemotherapy, RNAi-therapy
 Global reach, higher impact
Global reach, higher impact