Impact Factor
Theranostics 2019; 9(21):6080-6098. doi:10.7150/thno.36514 This issue Cite
Research Paper
Injectable hydrogels for the sustained delivery of a HER2-targeted antibody for preventing local relapse of HER2+ breast cancer after breast-conserving surgery
1. State Key Laboratory of Molecular Engineering of Polymers, Department of Macromolecular Science, Fudan University, Shanghai 200438, China
2. Department of Breast Surgery, Obstetrics and Gynecology Hospital, Fudan University, Shanghai 200011, China
* Authors contributed equally.
Abstract
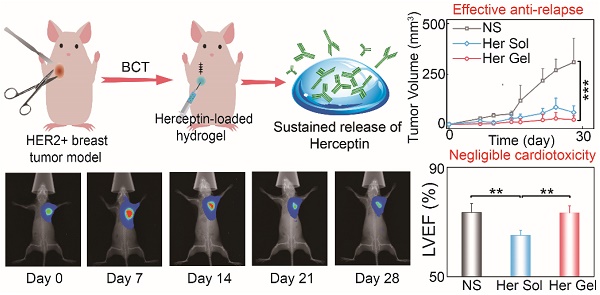
A high risk of local relapse is the main challenge of HER2+ breast cancer after breast-conserving surgery. We aimed to develop a long-acting delivery system for Herceptin, a HER2-targeting antibody, using injectable and thermosensitive hydrogels as the carrier to prevent the local relapse of HER2+ breast tumors while minimizing systemic side effects, especially cardiotoxicity.
Methods: Two poly(lactic acid-co-glycolic acid)-b-poly(ethylene glycol)-b-poly(lactic acid-co-glycolic acid) (PLGA-PEG-PLGA) triblock copolymers with different PEG/PLGA proportions were synthesized. Their mixtures with rational mix proportions displayed sol-gel transitions in water with rising of temperature and the Herceptin-loaded hydrogel systems were then prepared. Both the in vivo antitumor and anti-relapse efficacies were evaluated after hypodermic injection of the Herceptin-loaded hydrogel, and the cardiotoxicity was also detected.
Results: The gel performance, degradation rate and drug release kinetics of hydrogels were easily adjustable by simply varying the mix proportion. The hydrogel matrix with a specific mix proportion not only avoided initial burst release but also achieved sustained release of Herceptin in vitro for up to 80 days, which is the longest period of Herceptin delivery that has ever been reported. In vivo biodistribution studies performed in SK-BR-3 tumor-bearing mice revealed that a single hypodermic administration of the Herceptin-loaded hydrogel adjacent to the tumor tissue promoted the intratumoral antibody accumulation. This resulted in a better antitumor efficacy compared to weekly hypodermic injections of Herceptin solution for 28 days. A tumor relapse model was also established by imitative breast-conserving surgery on tumor-bearing mice, and both the single injection of the Herceptin-loaded hydrogel and the weekly injection of the Herceptin solution achieved superior anti-relapse efficacy. Furthermore, both antitumor and anti-relapse experiments demonstrated that the weekly pulsed administration of the Herceptin solution caused cardiotoxicity; however, the sustained release of Herceptin from the hydrogel effectively prevented this side effect.
Conclusion: The Herceptin-loaded hydrogel has great potential for preventing the relapse of HER2+ breast tumors after breast-conserving surgery with enhanced therapeutic efficacy, improved patient compliance and significantly reduced side effects.
Keywords: Injectable hydrogel, Breast-conserving surgery, Herceptin, Cardiotoxicity, Anti-relapse, Immunotherapy
 Global reach, higher impact
Global reach, higher impact