Impact Factor
Theranostics 2019; 9(21):6191-6208. doi:10.7150/thno.37538 This issue Cite
Research Paper
Kidney-targeted rhein-loaded liponanoparticles for diabetic nephropathy therapy via size control and enhancement of renal cellular uptake
1. Department of Ultrasound in Medicine, The Second Affiliated Hospital of Zhejiang University School of Medicine, Zhejiang University, Hangzhou 310009, China.
2. Department of Pediatrics, The First Affiliated Hospital of Zhejiang Chinese Medical University, Zhejiang Chinese Medical University, Hangzhou, 310006 China.
3. MOE Key Laboratory of Macromolecular Synthesis and Functionalization, Department of Polymer Science and Engineering, Zhejiang University, Hangzhou 310027, China.
Abstract
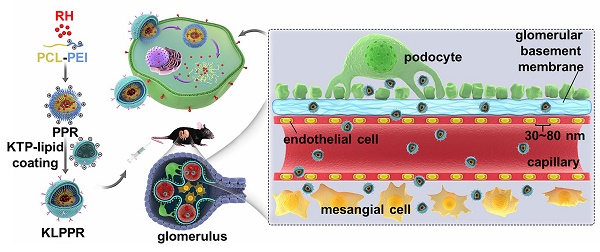
The optimization of nanoparticle size for passing through glomerular filtration membrane, inefficient renal cellular uptake and rapid urinary excretion of nanoparticles are the major obstacles for renal disease treatment via a nanoparticle delivery system. Herein, we propose a concept of a two-step nanoparticular cascade of size control and enhancement of renal cellular uptake to overcome the renal delivery obstacles.
Methods: We prepared kidney-targeted rhein (RH)-loaded liponanoparticles (KLPPR) with a yolk-shell structure composed by polycaprolactone-polyethyleneimine (PCL-PEI)-based cores and kidney targeting peptide (KTP)-modified lipid layers. The KLPPR size within the range of 30 ~ 80 nm allowed KLPPR distribute into kidney by passing through the glomerular filtration membrane and the KTP (sequence: CSAVPLC) decoration promoted the renal cellular uptake and endocytosis via a non-lysosomal pathway.
Results: The KLPPR had an average size of 59.5±6.2 nm and exhibited high RH loading, sustained release, good stability and biocompatibility, rapid cellular uptake in HK-2 cells. In addition, intravenous administration of KLPPR resulted in excellent kidney-targeted distribution and low urinary excretion in mice with streptozocin-induced diabetic nephropathy (DN), lowered the parameters of urea nitrogen, serum creatinine and kidney index, as well as facilitated the recovery of renal physiological function in improving the levels of urinary creatinine and the creatinine clearance rate by suppressing secretion and accumulation of fibronectin and TGF-β1.
Conclusion: Definitely, KLPPR were able to target the diseased kidney and improve the therapeutic effect of RH on DN by exploiting the two-step nanoparticular cascade of size control and enhancement of cellular uptake. This study offers a promising strategy for renal diseases treatment using liponanoparticle delivery system.
Keywords: diabetic nephropathy, rhein, liponanoparticles, size-control, kidney-targeted drug delivery
 Global reach, higher impact
Global reach, higher impact