Impact Factor
Theranostics 2019; 9(24):7184-7199. doi:10.7150/thno.38032 This issue Cite
Research Paper
Gene therapy for hereditary hearing loss by SLC26A4 mutations in mice reveals distinct functional roles of pendrin in normal hearing
1. Department of Biology, College of Natural Sciences, Kyungpook National University, Daegu 41566, Republic of Korea
2. School of Life Sciences, BK21 Plus KNU Creative BioResearch Group, Kyungpook National University, Daegu 41566, Republic of Korea
3. Department of Otorhinolaryngology, Head and Neck Surgery, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
4. Department of Anatomy, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
5. BK21PLUS Project for Medical Science, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
6. Department of Otolaryngology, College of Medicine, National Taiwan University, Taipei, Taiwan
7. Department of Otorhinolaryngology-Head and Neck Surgery, School of Medicine, Kyungpook National University, Daegu 41944, Republic of Korea
8. Department of Anatomy and Physiology, Kansas State University, Manhattan, United States of America
*These authors contributed equally to this work.
Abstract
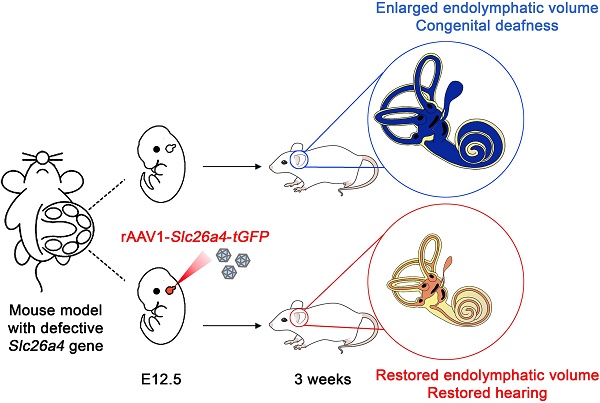
Rationale: Mutations of SLC26A4 that abrogate pendrin, expressed in endolymphatic sac, cochlea and vestibule, are known to cause autosomal recessive sensorineural hearing loss with enlargement of the membranous labyrinth. This is the first study to demonstrate the feasibility of gene therapy for pendrin-related hearing loss.
Methods: We used a recombinant viral vector to transfect Slc26a4 cDNA into embryonic day 12.5 otocysts of pendrin-deficient knock-out (Slc26a4∆/∆) and pendrin-deficient knock-in (Slc26a4tm1Dontuh/tm1Dontuh) mice.
Results: Local gene-delivery resulted in spatially and temporally limited pendrin expression, prevented enlargement, failed to restore vestibular function, but succeeded in the restoration of hearing. Restored hearing phenotypes included normal hearing as well as sudden, fluctuating, and progressive hearing loss.
Conclusion: Our study illustrates the feasibility of gene therapy for pendrin-related hearing loss, suggests differences in the requirement of pendrin between the cochlea and the vestibular labyrinth, and documents that insufficient pendrin expression during late embryonal and early postnatal development of the inner ear can cause sudden, fluctuating and progressive hearing loss without obligatory enlargement of the membranous labyrinth.
Keywords: Solute carrier family 26 member 4, Enlarged vestibular aqueduct, Recombinant adeno-associated virus, Gene therapy, In-utero, Pendred syndrome
 Global reach, higher impact
Global reach, higher impact