13.3
Impact Factor
Theranostics 2019; 9(24):7200-7209. doi:10.7150/thno.39831 This issue Cite
Research Paper
Cooperation of endogenous and exogenous reactive oxygen species induced by zinc peroxide nanoparticles to enhance oxidative stress-based cancer therapy
1. Department of Ultrasound Medicine, Laboratory of Ultrasound Molecular Imaging, the Third Affiliated Hospital of Guangzhou Medical University, Guangzhou 510150, China
2. Laboratory of Molecular Imaging and Nanomedicine (LOMIN), National Institute of Biomedical Imaging and Bioengineering (NIBIB), National Institutes of Health (NIH), Bethesda, Maryland 20892, United States
3. Department of Ultrasound, the First Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang 150076, China
4. MOE Key Laboratory for Analytical Science of Food Safety and Biology, College of Chemistry, Fuzhou University, Fuzhou 350108, China
5. Department of Radiology, the Fourth Hospital of Harbin Medical University, Harbin, Heilongjiang 150076, China
*These authors contributed equally to this work.
Abstract
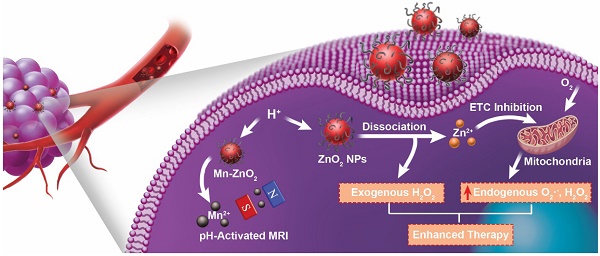
Reactive oxygen species (ROS)-generating anticancer agents can act through two different mechanisms: (i) elevation of endogenous ROS production in mitochondria, or (ii) formation/delivery of exogenous ROS within cells. However, there is a lack of research on the development of ROS-generating nanosystems that combine endogenous and exogenous ROS to enhance oxidative stress-mediated cancer cell death.
Methods: A ROS-generating agent based on polymer-modified zinc peroxide nanoparticles (ZnO2 NPs) was presented, which simultaneously delivered exogenous H2O2 and Zn2+ capable of amplifying endogenous ROS production for synergistic cancer therapy.
Results: After internalization into tumor cells, ZnO2 NPs underwent decomposition in response to mild acidic pH, resulting in controlled release of H2O2 and Zn2+. Intriguingly, Zn2+ could increase the production of mitochondrial O2·- and H2O2 by inhibiting the electron transport chain, and thus exerted anticancer effect in a synergistic manner with the exogenously released H2O2 to promote cancer cell killing. Furthermore, ZnO2 NPs were doped with manganese via cation exchange, making them an activatable magnetic resonance imaging contrast agent.
Conclusion: This study establishes a ZnO2-based theranostic nanoplatform which achieves enhanced oxidative damage to cancer cells by a two-pronged approach of combining endogenous and exogenous ROS.
Keywords: reactive oxygen species, zinc peroxide nanoparticles, pH-responsiveness, magnetic resonance imaging, cancer therapy
 Global reach, higher impact
Global reach, higher impact