Impact Factor
Theranostics 2019; 9(24):7268-7281. doi:10.7150/thno.33006 This issue Cite
Research Paper
Maf1 ameliorates cardiac hypertrophy by inhibiting RNA polymerase III through ERK1/2
1. Department of Cardiology, the First Affiliated Hospital of Sun Yat-Sen University, Guangzhou, Guangdong, China, 510080
2. Department of Cardiology, the Second People's Hospital of Guangdong Province, Guangzhou, Guangdong, China, 510080
3. NHC Key Laboratory of Assisted Circulation (Sun Yat-sen University), Guangzhou, China, 510080
4. Division of Cardiology, Department of Medicine, the University of Hong Kong Shenzhen Hospital, Shenzhen, Guangdong, China, 518000
5. Department of Cardiology, Center for Translational Medicine, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, China.
* Yu Sun, Cong Chen and Ruicong Xue contributed equally to the paper.
Abstract
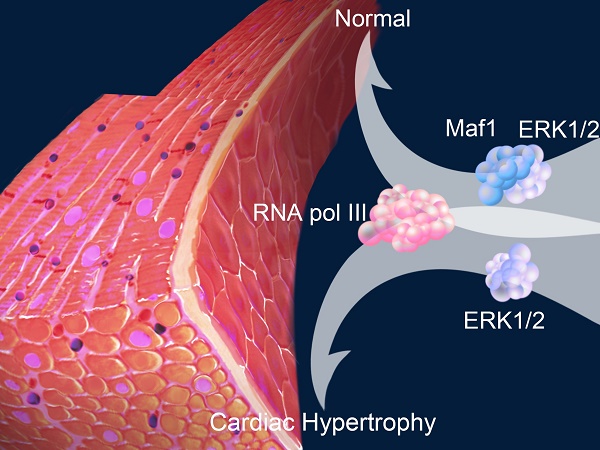
Rationale: An imbalance between protein synthesis and degradation is one of the mechanisms of cardiac hypertrophy. Increased transcription in cardiomyocytes can lead to excessive protein synthesis and cardiac hypertrophy. Maf1 is an RNA polymerase III (RNA pol III) inhibitor that plays a pivotal role in regulating transcription. However, whether Maf1 regulates of cardiac hypertrophy remains unclear.
Methods: Cardiac hypertrophy was induced in vivo by thoracic aortic banding (AB) surgery. Both the in vivo and in vitro gain- and loss-of-function experiments by Maf1 knockout (KO) mice and adenoviral transfection were used to verify the role of Maf1 in cardiac hypertrophy. RNA pol III and ERK1/2 inhibitor were utilized to identify the effects of RNA pol III and ERK1/2. The possible interaction between Maf1 and ERK1/2 was clarified by immunoprecipitation (IP) analysis.
Results: Four weeks after surgery, Maf1 KO mice exhibited significantly exacerbated AB-induced cardiac hypertrophy characterized by increased heart size, cardiomyocyte surface area, and atrial natriuretic peptide (ANP) expression and by exacerbated pulmonary edema. Also, the deficiency of Maf1 causes more severe cardiac dilation and dysfunction than wild type (WT) mice after pressure overload. In contrast, compared with adenoviral-GFP injected mice, mice injected with adenoviral-Maf1 showed significantly ameliorated AB-induced cardiac hypertrophy. In vitro study has demonstrated that Maf1 could significantly block phenylephrine (PE)-induced cardiomyocyte hypertrophy by inhibiting RNA pol III transcription. However, application of an RNA pol III inhibitor markedly improved Maf1 knockdown-promoted cardiac hypertrophy. Moreover, ERK1/2 was identified as a regulator of RNA pol III, and ERK1/2 inhibition by U0126 significantly repressed Maf1 knockdown-promoted cardiac hypertrophy accompanied by suppressed RNA pol III transcription. Additionally, IP analysis demonstrated that Maf1 could directly bind ERK1/2, suggesting Maf1 could interact with ERK1/2 and then inhibit RNA pol III transcription so as to attenuate the development of cardiac hypertrophy.
Conclusions: Maf1 ameliorates PE- and AB-induced cardiac hypertrophy by inhibiting RNA pol III transcription via ERK1/2 signaling suppression.
Keywords: Maf1, cardiac hypertrophy, RNA polymerase III, ERK1/2
 Global reach, higher impact
Global reach, higher impact