Impact Factor
Theranostics 2019; 9(24):7359-7369. doi:10.7150/thno.37625 This issue Cite
Research Paper
Carboxylesterase-Cleavable Biotinylated Nanoparticle for Tumor-Dual Targeted Imaging
1. Hefei National Laboratory of Physical Sciences at Microscale, Department of Chemistry, University of Science and Technology of China, 96 Jinzhai Road, Hefei, Anhui 230026, China
2. Department of PET Center, The First Affiliated Hospital, College of Medicine, Zhejiang University, 79 Qingchun Road, Hangzhou, Zhejiang 310003, China
3. Jiangsu Institute of Nuclear Medicine, 20 Qianrong Road, Wuxi, Jiangsu 214063, China
4. School of Life Sciences, University of Science and Technology of China, 443 Huangshan Road, Hefei, Anhui 230027, China
Abstract
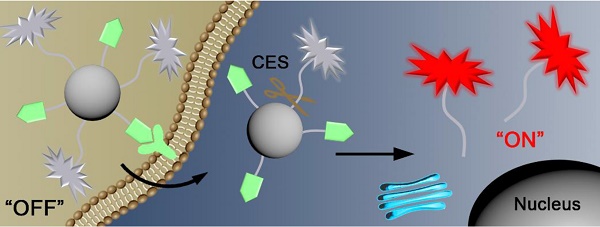
Near-infrared (NIR) nanoprobes with fluorescence “Turn-On” property are advantageous in cancer diagnosis but, to the best of our knowledge, “smart” nanoprobe that simultaneously targets both biotin receptor and carboxylesterase (CES) for HepG2 tumor-dual targeted imaging has not been reported.
Methods: Using CBT-Cys click condensation reaction, we rationally designed a “smart” NIR fluorescence probe H2N-Cys(StBu)-Lys(Biotin)-Ser(Cy5.5)-CBT (NIR-CBT) and used it to facilely prepare the fluorescence-quenched nanoparticle NIR-CBT-NP.
Results: In vitro results indicated that, after NIR-CBT-NP was incubated with CES for 6 h, its fluorescence was turned “On” by 69 folds. Cell experiments verified that NIR-CBT-NP was uptaken by HepG2 cells via biotin receptor-assisted endocytosis and its fluorescence was turned “On” by intracellular CES hydrolysis. Moreover, NIR-CBT-NP was successfully applied to image both biotin receptor- and CES-overexpressing HepG2 tumors.
Conclusion: Fluorescence-quenched nanoparticle NIR-CBT-NP was facilely prepared to actively target biotin receptor-overexpressing HepG2 cancer cells and turn the fluorescence “On” by intracellular CES hydrolysis for tumor-dual targeted imaging. We anticipate that our fluorescence “Turn-On” nanoparticle could be applied for liver cancer diagnosis in clinic in the near future.
Keywords: near-infrared, nanoparticle, carboxylesterase, biotin receptor, tumor imaging
 Global reach, higher impact
Global reach, higher impact