13.3
Impact Factor
Theranostics 2019; 9(24):7458-7473. doi:10.7150/thno.38081 This issue Cite
Research Paper
Orally Deliverable Nanotherapeutics for the Synergistic Treatment of Colitis-Associated Colorectal Cancer
1. Department of Medical Oncology; Sir Run Run Shaw Hospital; School of Medicine, Zhejiang University, Hangzhou, 310016, PR China.
2. The First Affiliated Hospital; Key Laboratory of Combined Multi-Organ Transplantation, Ministry of Public Health; School of Medicine, Zhejiang University, Hangzhou, 310003, PR China.
Abstract
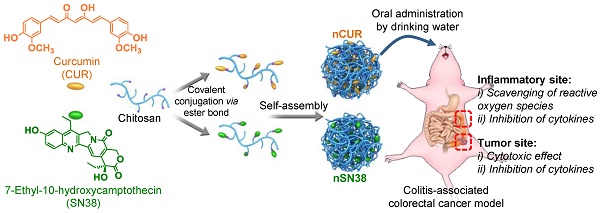
Purpose: Colitis-associated colorectal cancer (CAC) poses substantial challenges for effective treatment. Currently, there is a considerable need for the development of orally bioavailable dosage forms that enable the safe and effective delivery of therapeutic drugs to local diseased lesions in the gastrointestinal tract.
Experimental Design: In this study, we developed orally deliverable nanotherapeutics for the synergistic treatment of inflammatory bowel diseases (IBDs) and CAC. Water-insoluble curcumin (CUR) and 7-ethyl-10-hydroxycamptothecin (SN38), which served as anti-inflammatory and cytotoxic agents, respectively, were chemically engineered into hydrophilic mucoadhesive chitosan for the generation of chitosan-drug amphiphiles.
Results: The resulting amphiphilic constructs formed core-shell nanostructures in aqueous solutions and were orally administered for in vivo therapeutic studies. Using a preclinical CAC mouse model, we showed that the orally delivered nanotherapeutics locally accumulated in inflamed intestinal regions and tumor tissues. Furthermore, the therapeutic synergy of the combined nanotherapeutics in CAC mice was evaluated. Compared with their individual drug forms, combined CUR and SN38 nanoparticles yielded synergistic effects to alleviate intestinal inflammation and protect mice from ulcerative colitis. Notably, the combinatorial therapy demonstrated a remarkable tumor shrinkage with only ~6% of the total tumors exceeding 4 mm in diameter, whereas ~35% of tumors were observed to exceed a diameter of 4 mm in the saline-treated CAC mice. These data suggest a new and reliable approach for improving the treatment of IBD and CAC.
Conclusions: Our results showed that bioadhesive chitosan materials can be used to produce colloidal-stable nanotherapeutics that are suitable for oral delivery. Both nanotherapeutics exhibited substantial accumulation in inflamed intestinal regions and tumor tissues and showed good synergy for treating CAC, warranting further clinical translation.
Keywords: oral delivery, anti-inflammation, cytotoxic nanoparticle, self-assembly, colitis-associated colorectal cancer
 Global reach, higher impact
Global reach, higher impact