Impact Factor
Theranostics 2019; 9(25):7680-7696. doi:10.7150/thno.37220 This issue Cite
Research Paper
Blood TfR+ exosomes separated by a pH-responsive method deliver chemotherapeutics for tumor therapy
1. Institute for Translational Medicine, Qingdao University, Qingdao 266021, China.
2. College of Materials Science and Engineering, Qingdao University of Science and Technology, Qingdao 266042, China.
3. Tianjin Key Laboratory of Composite and Functional Materials, School of Materials Science and Engineering, Tianjin University, Tianjin 300072, China.
4. School of Clinical Medicine, Weifang Medical University, Weifang 261042, China.
Abstract
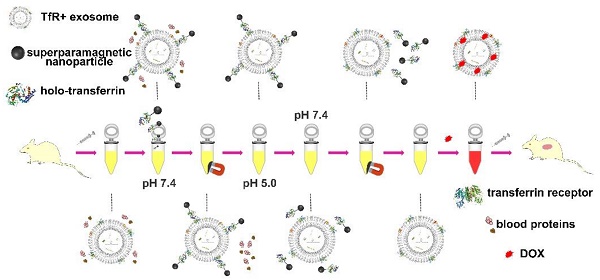
Blood transferrin receptor-positive (TfR+) exosomes are a kind of optimized drug delivery vector compared with other kinds of exosomes due to their easy access and high bio-safety. Their application facilitates the translation from bench to bedside of exosome-based delivery vehicles.
Methods: In this study, a pH-responsive superparamagnetic nanoparticles cluster (denoted as SMNC)-based method was developed for the precise and mild separation of blood TfR+ exosomes. Briefly, multiple superparamagnetic nanoparticles (SPMNs) labeled with transferrins (Tfs) could precisely bind to blood TfR+ exosomes to form an exosome-based cluster due to the specific recognition of TfR by Tf. They could realize the precise magnetic separation of blood TfR+ exosomes. More importantly, the pH-responsive dissociation characteristic of Tf and TfR led to the mild collapse of clusters to obtain pure blood TfR+ exosomes.
Results: Blood TfR+ exosomes with high purity and in their original state were successfully obtained through the pH-responsive SMNC-based method. These can load Doxorubicin (DOX) with a loading capacity of ~10% and dramatically increase the tumor accumulation of DOX in tumor-bearing mice because of their innate passive-targeting ability. In addition, blood TfR+ exosomes changed the biodistribution of DOX leading to the reduction of side effects. Compared with free DOX, DOX-loaded blood TfR+ exosomes showed much better tumor inhibition effects on tumor-bearing mice.
Conclusion: Taking advantage of the pH-responsive binding and disaggregation characteristics of Tf and TfR, the SMNC-based method can precisely separate blood TfR+ exosomes with high purity and in their original state. The resulting blood TfR+ exosomes showed excellent bio-safety and enable the efficient delivery of chemotherapeutics to tumors, facilitating the clinical translation of exosome-based drug delivery systems.
Keywords: blood TfR+ exosomes, superparamagnetic nanoparticles, pH-responsive, drug delivery, tumor therapy
 Global reach, higher impact
Global reach, higher impact