13.3
Impact Factor
Theranostics 2019; 9(25):7948-7960. doi:10.7150/thno.39203 This issue Cite
Research Paper
Integrin αvβ3-targeted radionuclide therapy combined with immune checkpoint blockade immunotherapy synergistically enhances anti-tumor efficacy
1. Department of Nuclear Medicine & Minnan PET Center, Xiamen Cancer Hospital, The First Affiliated Hospital of Xiamen University, China;
2. Department of Radiation Oncology, Xiamen Cancer Hospital, The First Affiliated Hospital of Xiamen University Teaching Hospital of Fujian Medical University, Xiamen, China;
3. State Key Laboratory of Molecular Vaccinology and Molecular Diagnostics & Center for Molecular Imaging and Translational Medicine, School of Public Health, Xiamen University, Xiamen, China;
4. Laboratory of Molecular Imaging and Nanomedicine, National Institute of Biomedical Imaging and Bioengineering, National Institutes of Health, Bethesda, Maryland, USA.
* These authors contributed equally to this work.
Abstract
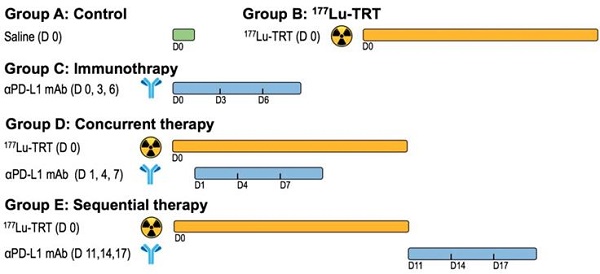
Rationale: Radiotherapy combined with immunotherapy has revealed promising outcomes in both preclinical studies and ongoing clinical trials. Targeted radionuclide therapy (TRT) is a branch of radiotherapy concerned with the use of radioisotopes, radiolabeled molecules or nanoparticles that deliver particulate radiation to cancer cells. TRT is a promising approach in cases of metastatic disease where conventional treatments are no longer effective. The increasing use of TRT raises the question of how to best integrate TRT with immunotherapy. In this study, we proposed a novel therapeutic regimen that combined programmed death ligand 1 (PD-L1)-based immunotherapy with peptide-based TRT (177Lu as the radionuclide) in the murine colon cancer model.
Methods: To explore the most appropriate timing of immunotherapy after radionuclide therapy, the anti-PD-L1 antibody (αPD-L1 mAb) was delivered in a concurrent or sequential manner when 177Lu TRT was given.
Results: The results demonstrated that TRT led to an acute increase in PD-L1 expression on T cells, and TRT in combination with αPD-L1 mAb stimulated the infiltration of CD8+ T cells, which improved local tumor control, overall survival and protection against tumor rechallenge. Moreover, our data revealed that the time window for this combination therapy may be critical to outcome.
Conclusions: This therapeutic combination may be a promising approach to treating metastatic tumors in which TRT can be used. Clinical translation of the result would suggest that concurrent rather than sequential blockade of the PD-1/PD-L1 axis combined with TRT improves overall survival and long-term tumor control.
Keywords: Targeted radionuclide therapy, immunotherapy, programed death ligand 1 (PD-L1), 177Lu-EB-RGD, combination treatment
 Global reach, higher impact
Global reach, higher impact