Impact Factor
Theranostics 2019; 9(26):8138-8154. doi:10.7150/thno.36762 This issue Cite
Research Paper
Bubble-Manipulated Local Drug Release from a Smart Thermosensitive Cerasome for Dual-Mode Imaging Guided Tumor Chemo-Photothermal Therapy
1. Department of Ultrasound, Peking University Third Hospital, Beijing, China
2. Ordos Center Hospital, Ordos 017000, Inner Mongolia, China
*These authors contributed equally
Abstract
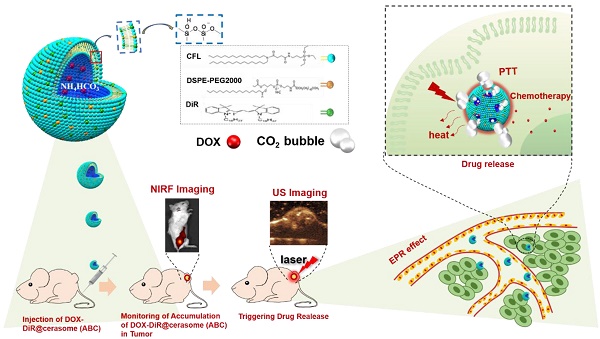
Thermosensitive liposomes have demonstrated great potential for tumor-specific chemotherapy. Near infrared (NIR) dyes loaded liposomes have also shown improved photothermal effect in cancer theranostics. However, the instability of liposomes often causes premature release of drugs or dyes, impeding their antitumor efficacy. Herein, we fabricated a highly stable thermo-responsive bubble-generating liposomal nanohybrid cerasome with a silicate framework, combined with a NIR dye to achieve NIR light stimulated, tumor-specific, chemo-photothermal synergistic therapy.
Methods: In this system, NIR dye of 1,1'-Dioctadecyl-3,3,3',3'- Tetramethylindotricarbocyanine iodide (DiR) with long carbon chains was self-assembled with a cerasome-forming lipid (CFL) to encapsulate ammonium bicarbonate (ABC), which was further used for actively loading doxorubicin (DOX), affording a thermosensitive and photosensitive DOX-DiR@cerasome (ABC).
Results: The resulting cerasome could disperse well in different media. Upon NIR light mediated thermal effect, ABC was decomposed to generate CO2 bubbles, resulting in a permeable channel in the cerasome bilayer that significantly enhanced DOX release. After intravenous injection into tumor-bearing mice, DOX-DiR@cerasome (ABC) could be efficiently accumulated at the tumor tissue, as monitored by DiR fluorescence, lasting for more than 5 days. NIR light irradiation was then performed at 36h to locally heat the tumors, resulting in immediate CO2 bubble generation, which could be clearly detected by ultrasound imaging, facilitating the monitoring process of controlled release of the drug. Significant antitumor efficacy could be obtained for the DOX-DiR@cerasome (ABC) + laser group, which was further confirmed by tumor tissue histological analysis.
Keywords: controlled drug release, combined therapy, cerasome, liposome, bubble responsive
 Global reach, higher impact
Global reach, higher impact