13.3
Impact Factor
Theranostics 2019; 9(26):8155-8170. doi:10.7150/thno.37023 This issue Cite
Research Paper
Proliferative regulation of alveolar epithelial type 2 progenitor cells by human Scnn1d gene
1. Department of Cellular and Molecular Biology, 11937 US Hwy 271, University of Texas Health Science Centre at Tyler, Tyler, Texas 75708-3154, USA.
2. Institute of Health Sciences, Key Laboratory of Medical Cell Biology of Ministry of Education,
3. Department of Stem Cells and Regenerative Medicine, College of Basic Medical Science, No. 77 Puhe Road, China Medical University, Shenyang North New Area, Shenyang, Liaoning Province 110122, China.
4. Barrow Neurological Institute, 350 West Thomas Road, St. Joseph's Hospital and Medical Centre, Phoenix, Arizona 85013-4409, USA.
5. Institute of Lung and Molecular Therapy, 601 Jinsui Avenue, Xinxiang Medical University, Xinxiang, Henan Province 453003, China.
6. Department of Pulmonary Immunology, 11937 US Hwy 271, University of Texas Health Science Centre at Tyler, Tyler, Texas 75708-3154, USA.
7. Department of Medicine and Women's Guild Lung Institute, 8700 Beverly Blvd., Suite 6724, Cedars-Sinai Medical Centre, Los Angeles, California 90048-3326, USA.
8. Department of Medicine and Anesthesia, 505 Parnassus Avenue, University of California San Francisco, San Francisco, California 94143-2204, USA.
9. Texas Lung Injury Institute, 11937 US Hwy 271, University of Texas Health Science Centre at Tyler, Tyler, Texas 75708-3154, USA.
Abstract
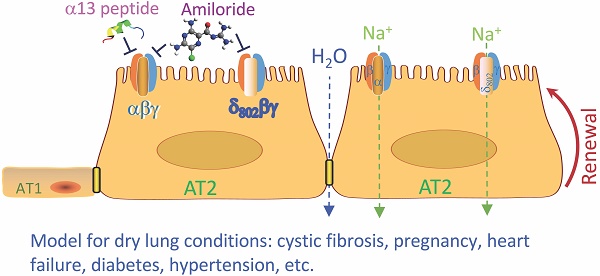
Lung epithelial sodium channel (ENaC) encoded by Scnn1 genes is essential for maintaining transepithelial salt and fluid homeostasis in the airway and the lung. Compared to α, β, and γ subunits, the role of respiratory δ-ENaC has not been studied in vivo due to the lack of animal models.
Methods: We characterized full-length human δ802-ENaC expressed in both Xenopus oocytes and humanized transgenic mice. AT2 proliferation and differentiation in 3D organoids were analysed with FACS and a confocal microscope. Both two-electrode voltage clamp and Ussing chamber systems were applied to digitize δ802-ENaC channel activity. Immunoblotting was utilized to analyse δ802-ENaC protein. Transcripts of individual ENaC subunits in human lung tissues were quantitated with qPCR.
Results: The results indicate that δ802-ENaC functions as an amiloride-inhibitable Na+ channel. Inhibitory peptide α-13 distinguishes δ802- from α-type ENaC channels. Modified proteolysis of γ-ENaC by plasmin and aprotinin did not alter the inhibition of amiloride and α-13 peptide. Expression of δ802-ENaC at the apical membrane of respiratory epithelium was detected with biophysical features similar to those of heterologously expressed channels in oocytes. δ802-ENaC regulated alveologenesis through facilitating the proliferation of alveolar type 2 epithelial cells.
Conclusion: The humanized mouse line conditionally expressing human δ802-ENaC is a novel model for studying the expression and function of this protein in vivo .
Keywords: alveolar type epithelial cells, epithelial sodium channels, self-renewal, humanized transgenic mouse line
 Global reach, higher impact
Global reach, higher impact