Impact Factor
Theranostics 2019; 9(26):8312-8320. doi:10.7150/thno.37128 This issue Cite
Research Paper
On-demand PEGylation and dePEGylation of PLA-based nanocarriers via amphiphilic mPEG-TK-Ce6 for nanoenabled cancer chemotherapy
1. School of Food and Biological Engineering, Hefei University of Technology, Hefei, Anhui, 230009, China
2. Institutes for Life Sciences, School of Medicine, South China University of Technology, Guangzhou, Guangdong 510006, China
3. Department of Laboratory Medicine, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, 510080, People's Republic of China
* These authors contributed equally to this work.
Abstract
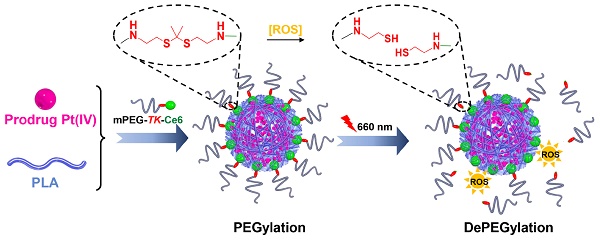
Rationale: PEGylation of nanocarriers could extend blood circulation time and enhance tumor accumulation via the enhanced permeability and retention (EPR) effect. Unfortunately, the PEG moiety suppresses tumor cell internalization of nanocarriers, resulting in limited therapeutic efficiency (known as the PEG dilemma). Designing stimuli-responsive shell-detachable nanocarriers, which could detach the PEG corona from the nanocarriers in desired tumor tissues in response to the local environment, is an appealing approach to overcome the PEG dilemma, but nanocarrier applications are also limited by a lack of universal stimuli for PEG detachment.
Methods: In this study, we synthesized red light-responsive, amphiphilic mPEG bridged to the photosensitizer Ce6 via a thioketal (TK) bond (mPEG-TK-Ce6), which was then used to achieve the PEGylation of polylactide (PLA)-based nanoparticles encapsulating the Pt(IV) prodrug. The therapeutic efficacy of the prepared nanoparticles was evaluated in vitro and in vivo.
Results: We demonstrated that the amphiphilic mPEG-TK-Ce6 can realize the PEGylation of Pt(IV) prodrug-loaded PLA nanoparticles and consequently enhanced nanoparticle accumulation in tumor tissues. When the tumor tissues were subjected to 660 nm irradiation, reactive oxygen species (ROS) generated by Ce6 induced the rapid degradation of the adjacent TK bond, resulting in PEG detachment and enhanced tumor cell internalization. Therefore, mPEG-TK-Ce6 facilely achieved PEGylation and light-responsive dePEGylation of the nanocarrier for enhanced antitumor efficacy in nanomedicine.
Conclusion: Such red light-responsive amphiphilic mPEG-TK-Ce6 facilely achieved PEGylation and dePEGylation of the nanocarrier, providing a facile strategy to overcome PEG dilemma.
Keywords: PEGylation, dePEGylation, photocleavable molecule, nanocarrier, cancer therapy
 Global reach, higher impact
Global reach, higher impact