Impact Factor
Theranostics 2019; 9(26):8332-8343. doi:10.7150/thno.35280 This issue Cite
Research Paper
Nanotopography-based lymphatic delivery for improved anti-tumor responses to checkpoint blockade immunotherapy
1. Center for Molecular Imaging, University of Texas Health Science Center, 3Department of Pathology and Laboratory Medicine, McGovern Medical School, Houston Texas 77030
2. Department of Pediatric Surgery, University of Texas Health Science Center, 3Department of Pathology and Laboratory Medicine, McGovern Medical School, Houston Texas 77030
3. Department of Melanoma Medical Oncology, M.D. Anderson Cancer Center, Houston, Texas 77030
4. Kimberly-Clark, Corporation, currently at Sorrento Therapeutics, San Diego, CA
5. Department of Pathology and Laboratory Medicine, McGovern Medical School, Houston Texas 77030
Abstract
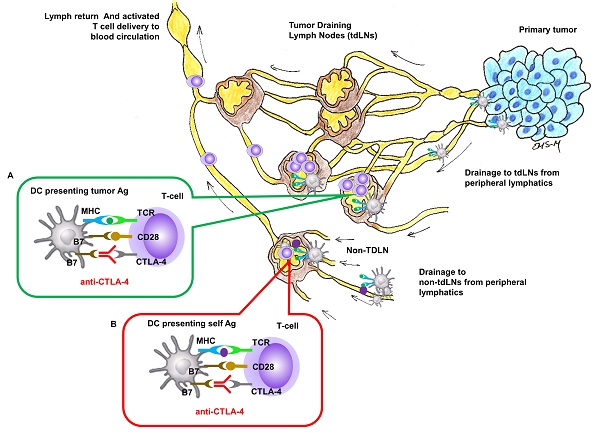
Rationale: Cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) is a co-inhibitory checkpoint receptor that is expressed by naïve T-cells in lymph nodes (LNs) to inhibit activation against “self” antigens (Ags). In cancer, anti-CTLA-4 blocks inhibitory action, enabling robust activation of T-cells against tumor Ags presented in tumor draining LNs (TDLNs). However, anti-CTLA-4 is administered intravenously with limited exposure within TDLNs and immune related adverse events (irAEs) are associated with over-stimulation of the immune system.
Methods: Herein, we first deliver anti-CTLA-4 in an orthotopic mammary carcinoma murine model using a nanotopographical microneedle-array device to compare its anti-tumor response to that from systemic administration. Additionally, to demonstrate the feasibility of lymphatic delivery in humans using the device, we use near-infrared fluorescence imaging to image delivery of ICG to LNs.
Results: Our data show that lymphatic infusion results in more effective tumor growth inhibition, arrest of metastases, increased tumor infiltrating lymphocytes and complete responses when compared to conventional systemic administration. In clinical studies, we demonstrate for the first time that nanotopographic infusion can deliver ICG through the lymphatics directly to the axilla and inguinal LNs of healthy human volunteers.
Conclusion: Taken together, these results suggest that regional delivery using a nanotopography-based microneedle array could revolutionize checkpoint blockade immunotherapy by reducing systemic drug exposure and maximizing drug delivery to TDLNs where tumor Ags present. Future work is needed to determine whether lymphatic delivery of anti-CTLA-4 can alleviate irAEs that occur with systemic dosing.
Keywords: Immunotherapy, Nanotopography, Lymphatic delivery, Fluorescence imaging
 Global reach, higher impact
Global reach, higher impact