Impact Factor
Theranostics 2019; 9(26):8362-8376. doi:10.7150/thno.39816 This issue Cite
Research Paper
Strategy to enhance lung cancer treatment by five essential elements: inhalation delivery, nanotechnology, tumor-receptor targeting, chemo- and gene therapy
1. Department of Pharmaceutics, Ernest Mario School of Pharmacy, Rutgers, the State University of New Jersey, Piscataway, NJ 08854, USA
2. Department of Pharmaceutical Sciences, College of Pharmacy, Oregon State University, Portland, OR 97201, USA
3. Rutgers Cancer Institute of New Jersey, New Brunswick, NJ 08903, USA
4. Departments of Pharmacology and Medicine, Rutgers, the State University of New Jersey, Piscataway, NJ 08854, USA
5. Environmental and Occupational Health Science Institute, Piscataway, NJ 08854, USA
Abstract
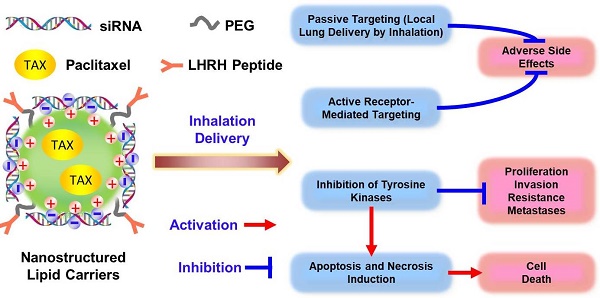
Non-Small Cell Lung Carcinoma (NSCLC), is the most common type of lung cancer (more than 80% of all cases). Small molecule Tyrosine Kinase (TK) Inhibitors acting on the Epidermal Growth Factor Receptors (EGFRs) are standard therapies for patients with NSCLC harboring EGFR-TK inhibitor-sensitizing mutations. However, fewer than 10 % of patients with NSCLC benefit from this therapy. Moreover, even the latest generation of EGFR inhibitors can cause severe systemic toxicities and are ineffective in preventing non-canonical EGFR signaling. In order to minimize and even overcome these limitations, we are proposing a novel multi-tier biotechnology treatment approach that includes: (1) suppression of all four types of EGFR-TKs by a pool of small interfering RNAs (siRNAs); (2) induction of cell death by an anticancer drug, (3) enhancing the efficiency of the treatment by the local inhalation delivery of therapeutic agents directly to the lungs (passive targeting), (4) active receptor-mediated targeting of the therapy specifically to cancer cells that in turn should minimize adverse side effects of treatment and (5) increasing the stability, solubility, and cellular penetration of siRNA and drug by using tumor targeted Nanostructured Lipid Carriers (NLC).
Methods: NLCs targeted to NSCLC cells by a synthetic Luteinizing Hormone-Releasing Hormone (LHRH) decapeptide was used for the simultaneous delivery of paclitaxel (TAX) and a pool of siRNAs targeted to the four major forms of EGFR-TKs. LHRH-NLC-siRNAs-TAX nanoparticles were synthesized, characterized and tested in vitro using human lung cancer cells with different sensitivities to gefitinib (inhibitor of EGFR) and in vivo on an orthotopic NSCLC mouse model.
Results: Proposed nanoparticle-based complex containing an anticancer drug, inhibitors of different types of EGFR-TKs and peptide targeted to the tumor-specific receptors (LHRH-NLC-siRNAs-TAX) demonstrated a favorable organ distribution and superior anticancer effect when compared with treatment by a single drug, inhibitor of one EGFR-TK and non-targeted therapy.
Conclusions: The use of a multifunctional NLC-based delivery system substantially enhanced the efficiency of therapy for NSCLC and possibly will limit adverse side effects of the treatments. The results obtained have the potential to significantly impact the field of drug delivery and to improve the efficiency of therapy of lung and other types of cancer.
Keywords: Imaging, LHRH peptide, suppression of EGFR-TK signaling pathways, pool of siRNAs, lung cancer cells with different resistance to gefitinib
 Global reach, higher impact
Global reach, higher impact