13.3
Impact Factor
Theranostics 2019; 9(26):8377-8391. doi:10.7150/thno.37628 This issue Cite
Research Paper
Inhibition of SMYD2 suppresses tumor progression by down-regulating microRNA-125b and attenuates multi-drug resistance in renal cell carcinoma
1. Department of Urology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China;
2. Institute of Urology of Hubei Province, Wuhan, China
3. Department of Urinary Surgery, First Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang, China.
4. Department of Urology, The First Affiliated Hospital of Xiamen University, Xiamen, China.
5. Department of Urology, The People's Hospital of Shenzhen City, Shenzhen, China.
6. Department of Molecular and Medical Pharmacology, David Geffen School of Medicine, University of California at Los Angeles, Los Angeles, USA
*These authors have contributed equally to this work
Abstract
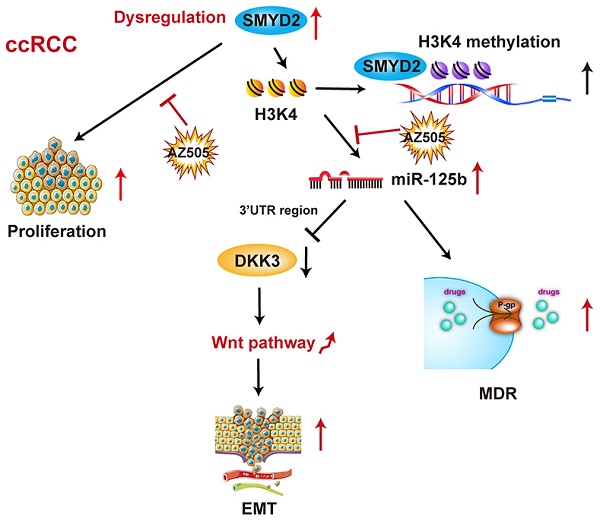
SMYD2 is a histone methyltransferase that has been reported to be an important epigenetic regulator. This study aims to investigate SMYD2 as a prognostic indicator of clear cell renal cell carcinoma (ccRCC) and explore its role in tumorigenesis and multi-drug resistance.
Methods: Tumor specimens, clinicopathologic information, and prognostic outcomes of 186 ccRCC patients from three hospitals in China were collected for SMYD2 immunohistochemistry staining, Kaplan-Meier analysis, and Cox proportional hazards-regression analysis. MicroRNA (miRNA)-microarray profiling identified differentially expressed miRNAs in renal cancer cells subjected to SMYD2 knockdown or treatment with the SMYD2 inhibitor AZ505. The effects of SMYD2 and candidate SMYD2-mediated miRNAs on renal cancer cell proliferation, migration, clonogenicity, and tumorigenicity were determined via cell-function assays and murine xenograft experiments. The half-inhibitory concentrations (IC50) of five antineoplastic drugs (cisplatin, doxorubicin, fluorouracil, docetaxel, and sunitinib) in AZ505-treated and control cells were calculated, and the effects of SMYD2 inhibition on P-glycoprotein (P-gP) expression and multiple-drug resistance were verified.
Results: SMYD2 was overexpressed and acted as an oncogene in ccRCC. High SMYD2 expression correlated with a high TNM stage (P = 0.007) and early tumor relapse (P = 0.032). SMYD2 independently predicted a worse overall survival (P = 0.022) and disease-free survival (P = 0.048). AZ505 inhibited the binding of SMYD2 to the miR-125b promoter region (based on chromatin immunoprecipitation assays) and suppressed ccRCC cell migration and invasion by inhibiting the SMYD2/miR-125b/DKK3 pathway. SMYD2 and miR-125b inhibition acted synergistically with anticancer drugs via P-gP suppression in vitro and in vivo.
Conclusions: These findings suggested that SMYD2 plays an important role in ccRCC development and could be a potential biomarker for the treatment and prognosis of RCC.
Keywords: SMYD2, Renal cell carcinoma, MicroRNA, Multidrug resistance
 Global reach, higher impact
Global reach, higher impact