Impact Factor
Theranostics 2020; 10(1):201-217. doi:10.7150/thno.35895 This issue Cite
Research Paper
Musashi-1 promotes stress-induced tumor progression through recruitment of AGO2
1. Division of Basic Research, Department of Medical Research, Taipei Veterans General Hospital, Taipei, Taiwan.
2. Institute of Clinical Medicine, School of Medicine, National Yang-Ming University, Taipei, Taiwan.
3. Institute of Pharmacology, School of Medicine, National Yang-Ming University, Taipei, Taiwan.
4. Institute of Food Safety and Health Risk Assessment, School of Pharmaceutical Sciences, National Yang-Ming University, Taipei, Taiwan.
5. Boston Children Hospital and Harvard Medical School, Boston MA, USA.
6. Department of Public Health, and Women's Hospital, Zhejiang University School of Medicine, Hangzhou, Zhejiang, China.
7. Division of Newborn Medicine and Epigenetics Program, Department of Medicine, Boston Children's Hospital, Boston, and Department of Cell Biology, Harvard Medical School, Boston, Massachusetts, USA.
8. Department of Neurosurgery, Taipei Veterans General Hospital, Taipei, Taiwan.
9. Department of Molecular and Cellular Oncology, University of Texas MD Anderson Cancer Center, Houston, Texas, USA.
10. Graduate Institute of Biomedical Sciences and Center for Molecular Medicine, China Medical University, Taichung, Taiwan.
11. Cancer Center, Taipei Veterans General Hospital, Taipei, Taiwan.
12. Department of Neurological Surgery, Tri-Service General Hospital and National Defense Medical Center, Taipei, Taiwan.
13. Genomic Research Center, Academia Sinica, Taipei, Taiwan.
14. Institute of Biomedical Sciences, Academia Sinica, Taipei, Taiwan.
* Equal contributions (co-first)
Abstract
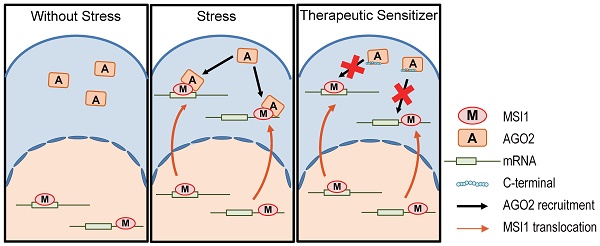
Carcinomatous progression and recurrence are the main therapeutic challenges frequently faced by patients with refractory tumors. However, the underlined molecular mechanism remains obscure.
Methods: We found Musashi-1 (MSI1) transported into cytosol under stress condition by confocal microscopy and cell fractionation. Argonaute 2 (AGO2) was then identified as a cytosolic binding partner of MSI1 by Mass Spectrametry, immunoprecipitation, and recombinant protein pull-down assay. We used RNA-IP to determine the MSI1/AGO2 associated regions on downstream target mRNAs. Finally, we overexpressed C-terminus of MSI1 to disrupt endogenous MSI1/AGO2 interaction and confirm it effects on tmor progression.
Results: Malignant tumors exhibit elevated level of cytosolic Musashi-1 (MSI1), which translocates into cytosol in response to stress and promote tumor progression. Cytosolic MSI1 forms a complex with AGO2 and stabilize or destabilize its target mRNAs by respectively binding to their 3´ untranslated region or coding domain sequence. Both MSI1 translocation and MSI1/AGO2 binding are essential for promoting tumor progression. Blocking MSI1 shuttling by either chemical inhibition or point mutation attenuates the growth of GBM-xenografts in mice. Importantly, overexpression of the C-terminus of MSI1 disrupts endogenous MSI1/AGO2 interaction and effectively reduces stress-induced tumor progression.
Conclusion: Our findings highlight novel molecular functions of MSI1 during stress-induced carcinomatous recurrence, and suggest a new therapeutic strategy for refractory malignancies by targeting MSI1 translocation and its interaction with AGOs.
Keywords: Musashi-1, Argonaute 2, RNA regulation, subcellular translocation, cancer, tumor recurrence
 Global reach, higher impact
Global reach, higher impact