Impact Factor
Theranostics 2020; 10(1):231-246. doi:10.7150/thno.37142 This issue Cite
Research Paper
Soluble CD146, a cerebrospinal fluid marker for neuroinflammation, promotes blood-brain barrier dysfunction
1. Savaid Medical School, University of Chinese Academy of Sciences, Beijing, China.
2. Key Laboratory of Protein and Peptide Pharmaceutical, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.
3. Neuroimmunology Laboratory, Peking University First Hospital, Beijing, China.
4. School of Basic Medical Sciences, Southwest Medical University, Sichuan, China.
5. Beijing Anzhen Hospital, Capital Medical University, Beijing, China.
* Contributed equally as first authors
† Contributed equally as last authors
Abstract
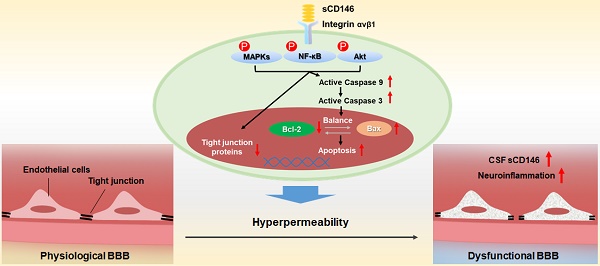
The blood-brain barrier (BBB) dysfunction is an initial event of various neuroinflammatory diseases. However, the absence of reliable markers and mechanisms for BBB damage greatly limits the diagnosis and treatment of neuroinflammatory diseases. Soluble CD146 (sCD146) is mainly derived from vascular endothelial cells (ECs) and highly elevated in inflammatory settings. Based on a small cohort, our previous study showed that sCD146 is elevated in the cerebrospinal fluid (CSF) of multiple sclerosis (MS), which is accompanied with BBB damage. Nevertheless, whether sCD146 monitors and regulates the BBB dysfunction remains unknown.
Methods: Coupled serum and CSF samples from patients with or without neuroinflammatory diseases were collected via multicenter collaborations. sCD146 was measured by sandwich ELISA using anti-CD146 antibodies AA1 and AA98, both of which were generated in our laboratory. The correlations between sCD146 and other clinical parameters or inflammatory factors were analyzed by Spearman's correlation coefficient analysis. The role of sCD146 on BBB function was examined in an in vitro BBB model.
Results: Between July 20, 2011, and February 31, 2017, we collected coupled serum and CSF samples from 823 patients, of which 562 (68.3%) had neuroinflammatory diseases, 44 (5.3%) had remitting MS, and 217 (26.4%) had non-inflammatory neurological diseases (NIND). We found that sCD146 in CSF, but not in serum, is abnormally elevated in neuroinflammatory diseases (37.3 ± 13.3 ng/mL) compared with NIND (4.7 ± 2.9 ng/mL) and remitting MS (4.6 ± 3.5 ng/mL). Abnormally elevated CSF sCD146 is significantly correlated with the hyperpermeability-related clinical parameters of BBB and neuroinflammation-related factors. Moreover, CSF sCD146 shows higher sensitivity and specificity for evaluating BBB damage. Using an in vitro BBB model, we found that sCD146 impairs BBB function by promoting BBB permeability via an association with integrin αvβ1. Blocking integrin αvβ1 significantly attenuates sCD146-induced hyperpermeability of the BBB.
Conclusion: Our study provides convincing evidence that CSF sCD146 is a sensitive marker of BBB damage and neuroinflammation. Furthermore, sCD146 is actively involved in BBB dysfunction.
Keywords: sCD146, cerebrospinal fluid, blood-brain barrier damage and neuroinflammation.
 Global reach, higher impact
Global reach, higher impact