13.3
Impact Factor
Theranostics 2020; 10(1):300-311. doi:10.7150/thno.38736 This issue Cite
Research Paper
Metastasis-on-a-chip mimicking the progression of kidney cancer in the liver for predicting treatment efficacy
1. State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, National Clinical Research Center for Infectious Diseases, Collaborative Innovation Center for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, 310003, China.
2. Institute for Translational Medicine, Zhejiang University, Hangzhou, Zhejiang Province, 310029, China.
3. Department of Bioengineering, University of California Berkeley, Berkeley, CA, 94720, United States of America.
4. State Key Laboratory of Fluid Power and Mechatronic Systems, Key Laboratory of 3D Printing Process and Equipment of Zhejiang Province College of Mechanical Engineering, Zhejiang University, Hangzhou, Zhejiang Province, 310029, China.
5. Wellcome Trust-Medical Research Council Cambridge Stem Cell Institute and Department of Clinical Neurosciences, University of Cambridge, Cambridge CB2 0AH, United Kingdom.
6. Department of Biomedical Engineering, The Hong Kong Polytechnic University, Hong Kong SAR, China.
7. Department of Pharmaceutical Science, Åbo Akademic University, FI-20520, Turku, Finland.
Abstract
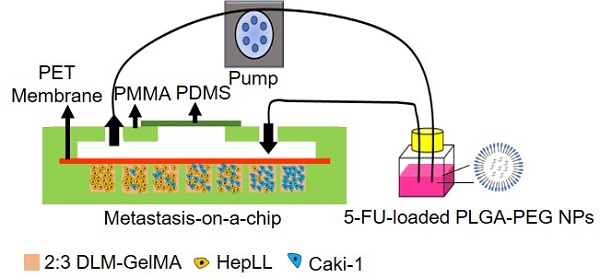
Metastasis is one of the most important factors that lead to poor prognosis in cancer patients, and effective suppression of the growth of primary cancer cells in a metastatic site is paramount in averting cancer progression. However, there is a lack of biomimetic three-dimensional (3D) in vitro models that can closely mimic the continuous growth of metastatic cancer cells in an organ-specific extracellular microenvironment (ECM) for assessing effective therapeutic strategies.
Methods: In this metastatic tumor progression model, kidney cancer cells (Caki-1) and hepatocytes (i.e., HepLL cells) were co-cultured at an increasing ratio from 1:9 to 9:1 in a decellularized liver matrix (DLM)/gelatin methacryloyl (GelMA)-based biomimetic liver microtissue in a microfluidic device.
Results: Via this model, we successfully demonstrated a linear anti-cancer relationship between the concentration of anti-cancer drug 5-Fluorouracil (5-FU) and the percentage of Caki-1 cells in the co-culture system (R2 = 0.89). Furthermore, the Poly(lactide-co-glycolide) (PLGA)-poly(ethylene glycol) (PEG)-based delivery system showed superior efficacy to free 5-FU in killing Caki-1 cells.
Conclusions: In this study, we present a novel 3D metastasis-on-a-chip model mimicking the progression of kidney cancer cells metastasized to the liver for predicting treatment efficacy. Taken together, our study proved that the tumor progression model based on metastasis-on-a-chip with organ-specific ECM would provide a valuable tool for rapidly assessing treatment regimens and developing new chemotherapeutic agents.
Keywords: Metastasis-on-a-chip, Tumor progression, Chemotherapy, Drug delivery.
 Global reach, higher impact
Global reach, higher impact