Impact Factor
Theranostics 2020; 10(1):323-339. doi:10.7150/thno.38870 This issue Cite
Research Paper
Fusobacterium nucleatum Promotes Metastasis in Colorectal Cancer by Activating Autophagy Signaling via the Upregulation of CARD3 Expression
1. Department of Gastroenterology, Renmin Hospital of Wuhan University, Wuhan, Hubei Province, China.
2. Key Laboratory of Hubei Province for Digestive System Disease, Wuhan, Hubei Province, China.
3. Central Laboratory, Renmin Hospital of Wuhan University, Wuhan, Hubei Province, China.
4. Department of Gastroenterology, The Affiliated Hospitalof North Sichuan Medical College, Road Wenhua 63#, Region Shunqing, Nanchong City 637000, China.
5. Center for Epigenetics & Disease Prevention, Institute of Biosciences and Technology, Texas A&M University, Houston, TX77030, USA.
# These authors contributed equally to this work.
Abstract
Aims: We aimed to measure the abundance of Fusobacterium nucleatum (F. nucleatum) in colorectal cancer (CRC) tissues from patients and to uncover the function of this bacterium in colorectal tumor metastasis.
Methods: We collected metastatic and non-metastatic CRC tissues to analyze F. nucleatum abundance. Cells were incubated with F. nucleatum or chloroquine (CQ) or were transfected with CARD3-targeting siRNA; the expression of mRNAs and proteins was then measured. CRC cells stably transfected with shRNA-luc were mixed with F. nucleatum and intravenously injected into BALB/cJ mice. APCMin/+, CARD3-/-and CARD3wt C57BL mice were given F. nucleatum; some mice were given azoxymethane (AOM) and dextran sodium sulfate (DSS).
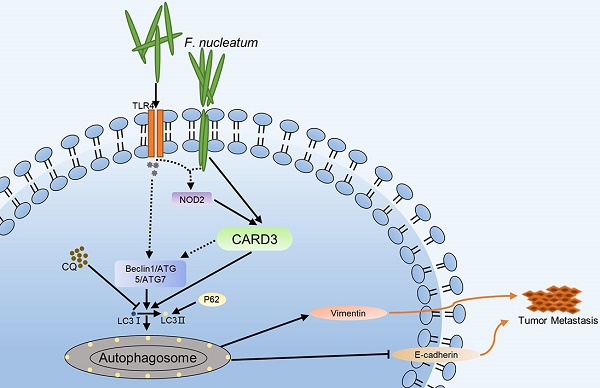
Results: F. nucleatum was abundant in CRC tissues from patients with metastasis. F. nucleatum infection increased CRC cell motility and upregulated the expression of CARD3, LC3-II, Beclin1 and Vimentin, and downregulated the expression of E-cadherin and P62 in CRC cells. These effects were attenuated by treatment with CQ, siCARD3 or both. APCMin/+ mice gavaged with F. nucleatum developed more aggressive tumors than control mice. After AOM/DSS administration, the colorectums of CARD3-/- mice had fewer tumors than those of control mice. Tumors from CARD3-/- mice had lower levels of LC3-II and Beclin1 and higher levels of P62 than those from control mice. BALB/cJ mice injected with both CT26-luc cells and F. nucleatum formed more metastases than control mice. CQ treatment, CARD3 knockdown or both reduced the ability of CT26-luc cells to form metastases in vivo.
Conclusions: F. nucleatum is enriched in CRC tissues from patients with metastasis. F. nucleatum orchestrates CARD3 and autophagy to control CRC metastasis. Measuring and targeting F. nucleatum and its associated pathways will yield approaches for the prevention and treatment of CRC metastasis.
Keywords: microbe, gene regulation, gene targeting, colorectal cancer
 Global reach, higher impact
Global reach, higher impact