Impact Factor
Theranostics 2020; 10(1):340-352. doi:10.7150/thno.39066 This issue Cite
Research Paper
Stem Cell Factor-Inducible MITF-M Expression in Therapeutics for Acquired Skin Hyperpigmentation
1. College of Pharmacy, Chungbuk National University, Cheongju 28160, Korea.
2. College of Pharmacy, Chonnam National University, Gwangju 61186, Korea.
3. College of Pharmacy, Chungnam National University, Daejeon 34134, Korea.
*C.-Y.Y. and E.R. equally contributed to this study.
Abstract
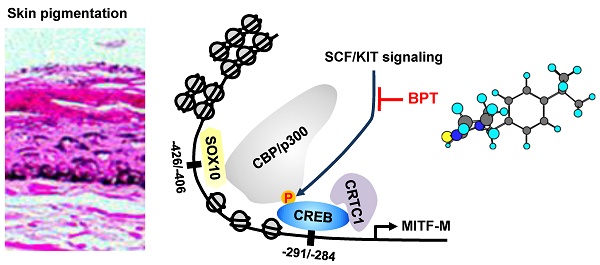
Rationale: Microphthalmia-associated transcription factor M (MITF-M) plays important roles in the pigment production, differentiation and survival of melanocytes. Stem cell factor (SCF) and its receptor KIT stimulate MITF-M activity via phosphorylation at the post-translation level. However, the phosphorylation shortens half-life of MITF-M protein over the course of minutes. Here, we investigated novel hypotheses of (i) whether SCF/KIT can regulate MITF-M activity through gene expression as the alternative process, and (ii) whether chemical inhibition of KIT activity can mitigate the acquired pigmentation in skin by targeting the expression of MITF-M.
Methods: We employed melanocyte cultures in vitro and pigmented skin samples in vivo, and applied immunoblotting, RT-PCR, siRNA-based gene knockdown and confocal microscopy.
Results: The protein and mRNA levels of MITF-M in epidermal melanocytes and the promoter activity of MITF-M in B16-F0 melanoma cells demonstrated that SCF/KIT could trigger the expression of MITF-M de novo, following the phosphorylation-dependent proteolysis of pre-existing MITF-M protein. SCF/KIT regulated the transcription abilities of cAMP-responsive element-binding protein (CREB), CREB-regulated co-activator 1 (CRTC1) and SRY-related HMG-box 10 (SOX10) but not β-catenin at the MITF-M promoter. Meanwhile, chemical inhibition of KIT activity abolished SCF-induced melanin production in epidermal melanocyte cultures, as well as protected the skin from UV-B-induced hyperpigmentation in HRM2 mice or brownish guinea pigs, in which it down-regulated the expression of MITF-M de novo at the promoter level.
Conclusion: We propose the targeting of SCF/KIT-inducible MITF-M expression as a strategy in the therapeutics for acquired pigmentary disorders.
Keywords: Stem cell factor, KIT, MITF-M activity, epidermal melanocyte, skin pigmentation, chemical inhibition
 Global reach, higher impact
Global reach, higher impact