Impact Factor
Theranostics 2020; 10(1):371-383. doi:10.7150/thno.39412 This issue Cite
Research Paper
A cancer-specific activatable theranostic nanodrug for enhanced therapeutic efficacy via amplification of oxidative stress
State Key Laboratory of Natural Medicines, Jiangsu Key Laboratory of TCM Evaluation and Translational Research, Research Center for Traceability and Standardization of TCMs, Cellular and Molecular Biology Center, School of Traditional Chinese Pharmacy, China Pharmaceutical University, Nanjing 211198, P.R. China.
* These authors contributed equally to this work.
Abstract
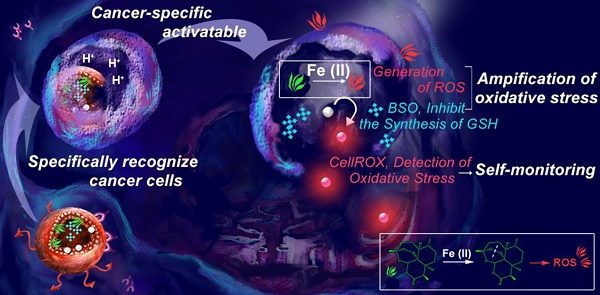
Rationale: Despite considerable advances, the reactive oxygen species (ROS)-mediated cancer treatment suffers from the problems of up-regulation of adaptive antioxidants in cancer cells as well as side effects to normal cells. Therefore, development of a new generation of cancer-specific nanomedicine capable of amplifying oxidative stress would be of great interest for accurate and effective cancer treatment.
Methods: Herein, transferrin (Tf)-decorated, dihydroartemisinin (DHA), L-buthionine-sulfoximine (BSO), and CellROX-loaded liposomal nanoparticles (Tf-DBC NPs) were developed for precise cancer theranositcs. Tf-DBC NPs could specifically recognize cancer cells via Tf-Tf receptor binding and be uptaken into the lysosomes of cancer cells, where Tf-DBC NPs were activated to release Fe(II), DHA, and BSO. ROS was generated by DHA in the presence of Fe(II), and GSH was depleted by BSO to disrupt the redox balance in cancer cells. Furthermore, CellROX, as a fluorescent probe for imaging of intracellular oxidative stress, was used to monitor the therapeutic efficacy.
Results: The integration of Tf, DHA, and BSO into the acidic pH-responsive liposomes selectively and effectively killed cancer cells and prevented the oxidative injury to normal cells. The high oxidative state was visualized at the tumor site and the amplification of oxidative stress enabled tumor eradication by Tf-DBC NPs, demonstrating the successful implementation of this novel strategy in vivo.
Conclusion: Our study provides a new paradigm for the design of ROS-mediated therapeutics and offers a promising perspective for precise cancer treatment.
Keywords: cancer, amplification of oxidative stress, theranostics, dihydroartemisinin, nanodrug
 Global reach, higher impact
Global reach, higher impact