13.3
Impact Factor
Theranostics 2020; 10(1):384-397. doi:10.7150/thno.40098 This issue Cite
Research Paper
Bax inhibitor 1 preserves mitochondrial homeostasis in acute kidney injury through promoting mitochondrial retention of PHB2
1. Chinese PLA General Hospital, Medical School of Chinese PLA, Beijing, 100853, China.
2. Center for Cardiovascular Research and Alternative Medicine, University of Wyoming College of Health Sciences, Laramie, WY 82071 USA.
3. Department of Cardiology and Shanghai Institute of Cardiovascular Diseases, Zhongshan Hospital, Fudan University, Shanghai, China 200032.
*The first two authors contributed equally to this article.
Abstract
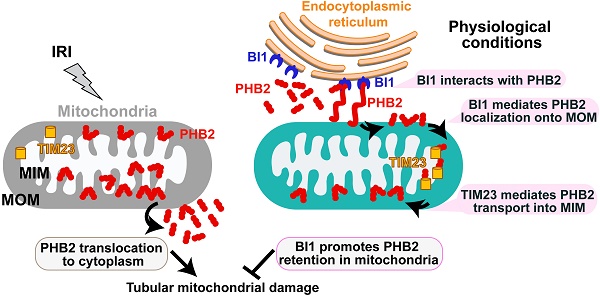
Bax inhibitor-1 (BI1) conveys anti-apoptotic signals for mitochondria while prohibitin 2 (PHB2) is implicated in sustaining mitochondrial morphology and function. However, their regulatory roles in acute kidney injury (AKI) are largely unknown.
Methods: In human patients with AKI, levels of BI1 in urine and plasma were determined using ELISA. An experimental model of AKI was established using ATP depletion-mediated metabolic stress and ischemia-reperfusion injury (IRI) in primary tubule cells and BI1 transgenic mice, respectively. Western blots, ELISA, qPCR, immunofluorescence, RNA silencing, and domain deletion assay were employed to evaluate the roles of BI1 and PHB2 in the preservation of mitochondrial integrity.
Results: Levels of BI1 in urine and plasma were decreased in patients with AKI and its expression correlated inversely with renal function. However, reconstitution of BI1 in a murine AKI model was capable of alleviating renal failure, inflammation and tubular death. Further molecular scrutiny revealed that BI1 preserved mitochondrial genetic integrity, reduced mitochondrial oxidative stress, promoted mitochondrial respiration, inhibited excessive mitochondrial fission, improved mitophagy and suppressed mitochondrial apoptosis. Intriguingly, levels of the mitochondria-localized PHB2 were sustained by BI1 and knockdown of PHB2 abolished the mitochondrial- and renal- protective properties of BI1. Furthermore, BI1 promoted PHB2 retention within mitochondria through direct interaction with cytoplasmic PHB2 to facilitate its mitochondrial import. This was confirmed by the observation that the C-terminus of BI1 and the PHB domain of PHB2 were required for the BI1-PHB2 cross-linking.
Conclusion: Our data have unveiled an essential role of BI1 as a master regulator of renal tubule function through sustaining mitochondrial localization of PHB2, revealing novel therapeutic promises against AKI.
Keywords: BI1, mitochondria, tubule cells, AKI, PHB2.
 Global reach, higher impact
Global reach, higher impact