13.3
Impact Factor
Theranostics 2020; 10(2):553-566. doi:10.7150/thno.37181 This issue Cite
Research Paper
NFATc3-dependent expression of miR-153-3p promotes mitochondrial fragmentation in cardiac hypertrophy by impairing mitofusin-1 expression
1. Center for Developmental Cardiology, Institute for Translational Medicine, College of Medicine, Qingdao University, Qingdao 266021, China
2. State Key Laboratory of Cardiovascular Disease, Heart Failure Center, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences, Peking Union Medical College, Beijing, 100037, China
# These authors contributed equally to this work
Abstract
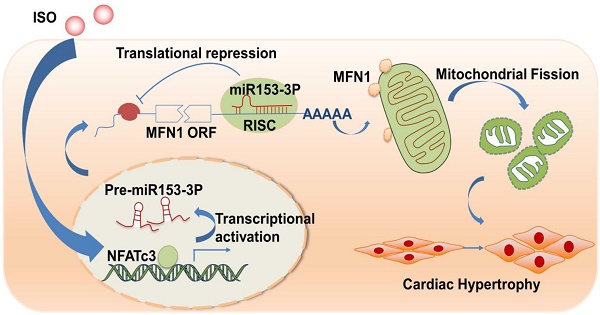
Mitochondrial dysfunction is involved in the pathogenesis of various cardiovascular disorders. Although mitochondrial dynamics, including changes in mitochondrial fission and fusion, have been implicated in the development of cardiac hypertrophy, the underlying molecular mechanisms remain mostly unknown. Here, we show that NFATc3, miR-153-3p, and mitofusion-1 (Mfn1) constitute a signaling axis that mediates mitochondrial fragmentation and cardiomyocyte hypertrophy.
Methods: Isoprenaline (ISO) was used to stimulate the hypertrophic response and mitochondrial fragmentation in cultured cardiomyocytes and in vivo. We performed immunoblotting, immunofluorescence, and quantitative real-time PCR to validate the function of Mfn1 in cardiomyocyte hypertrophy. Bioinformatic analyses, a luciferase reporter assay, and gain- and loss-of-function studies were used to demonstrate the biological function of miR-153-3p, which regulates mitochondrial fragmentation and hypertrophy by targeting Mfn1. Moreover, ChIP-qPCR and a luciferase reporter assay were performed to identify transcription factor NFATc3 as an upstream regulator to control the expression of miR-153-3p.
Results: Our results show that ISO promoted mitochondrial fission and enhanced the expression of miR-153-3p in cardiomyocytes. Knockdown of miR-153-3p attenuated ISO-induced mitochondrial fission and hypertrophy in cultured primary cardiomyocytes. miR-153-3p suppression inhibited mitochondrial fragmentation in ISO-induced cardiac hypertrophy in a mouse model. We identified direct targeting of Mfn1, a key protein of the mitochondrial fusion process, by miR-153-3p. Also, miR-153-3p promoted ISO-induced mitochondrial fission by suppressing the translation of Mfn1. We further found that NFATc3 activated miR-153-3p expression. Knockdown of NFATc3 inhibited miR-153-3p expression and blocked mitochondrial fission and hypertrophic response in cardiomyocytes.
Conclusions: Our data revealed a novel signaling pathway, involving NFATc3, miR-153-3p, and Mfn1, which could be a therapeutic target for the prevention and treatment of cardiac hypertrophy.
Keywords: miR-153-3p, mitofusion-1, NFATc3, mitochondrial fragmentation, cardiac hypertrophy
 Global reach, higher impact
Global reach, higher impact