Impact Factor
Theranostics 2020; 10(2):567-584. doi:10.7150/thno.36777 This issue Cite
Research Paper
Delivery of polymeric nanostars for molecular imaging and endoradiotherapy through the enhanced permeability and retention (EPR) effect
1. Department of Radiology, Memorial Sloan-Kettering Cancer Center, New York, USA.
2. ARC Centre of Excellence in Convergent Bio-Nano Science & Technology, Monash Institute of Pharmaceutical Sciences, Monash University, Parkville, Australia.
3. Department of Biochemistry & Structural Biology, Weill Cornell Graduate School, New York, USA.
4. Weill Cornell/Rockefeller/Sloan Kettering Tri-Institutional MD-PhD Program, New York, USA.
5. Probing Biosystems Future Science Platform, Commonwealth Scientific and Industrial Research Organisation, Herston, Australia.
6. Nanoscale Organisation and Dynamics Group, Western Sydney University, Penrith, Australia.
7. Department of Radiology, the Molecular Pharmacology Program and the Radiochemistry and Molecular Imaging Probes Core, Memorial Sloan Kettering Cancer Center, New York, USA.
8. Departments of Radiology and Pharmacology, Weill Cornell Medical College, New York, USA.
9. Australian Institute for Bioengineering and Nanotechnology, University of Queensland, St Lucia, Australia.
Abstract
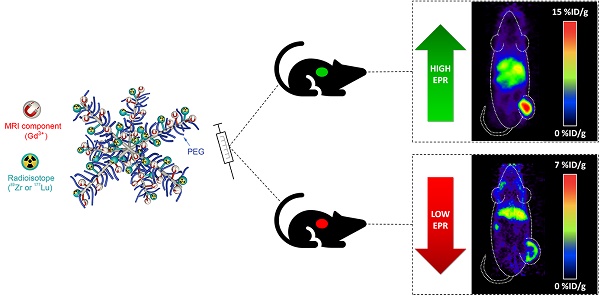
Expression levels of biomarkers are generally unknown at initial diagnosis. The development of theranostic probes that do not rely on biomarker availability would expand therapy options for cancer patients, improve patient selection for nanomedicine and facilitate treatment of inoperable patients or patients with acquired therapy resistance. Herein, we report the development of star polymers, also known as nanostars, that allow for molecular imaging and/or endoradiotherapy based on passive targeting via the enhanced permeability and retention (EPR) effect.
Methods: We synthesised a star copolymer, consisting of 7-8 centre-cross-linked arms that were modified with Gd3+ for magnetic resonance imaging (MRI), and functionalised either with 89Zr for in vivo quantification and positron emission tomography (PET) imaging, or with 177Lu for endoradiotherapy. 1H longitudinal relaxivities were determined over a continuum of magnetic field strengths ranging from 0.24 mT - 0.94 T at 37 °C (nuclear magnetic relaxation dispersion (NMRD) profile) and T1-weighted MRI contrast enhancement was visualized at 3 T and 7 T. PET imaging and ex vivo biodistribution studies were performed in mice bearing tumours with high EPR (CT26) or low EPR (BxPC3) characteristics. Therapy studies were performed in mice with high EPR tumours and mean absorbed organ doses were estimated for a standard human model.
Results: The star copolymer with Gd3+ displayed a significantly superior contrast enhancement ability (T1 = 0.60 s) compared to the standard clinical contrast agent Gadovist (T1 = 1.0 s). Quantification of tumour accumulation using the radiolabelled nanostars in tumour-bearing mice demonstrated an exceptionally high uptake in tumours with high EPR characteristics (14.8 - 21.7 %ID/g). Uptake of the star polymers in tumours with low EPR characteristics was significantly lower (P<0.001), suggesting passive tumour accumulation of the nanostars via the EPR effect. Survival of mice treated with high dose 177Lu-labelled star polymers was significantly higher than survival of mice treated with lower therapy doses or control mice (P=0.001), demonstrating the utility of the 177Lu-labelled star polymers as platforms for endoradiotherapy.
Conclusion: Our work highlights the potential of star polymers as probes for the molecular imaging of cancer tissue or for the passive delivery of radionuclides for endoradiotherapy. Their high functionalisability and high tumour accumulation emphasises their versatility as powerful tools for nanomedicine.
Keywords: star polymer, nanoparticle, EPR effect, imaging, therapy
 Global reach, higher impact
Global reach, higher impact