Impact Factor
Theranostics 2020; 10(2):602-614. doi:10.7150/thno.36220 This issue Cite
Research Paper
Mdig promotes oncogenic gene expression through antagonizing repressive histone methylation markers
1. Department of Pharmaceutical Sciences, Eugene Applebaum College of Pharmacy and Health Sciences, Wayne State University, 259 Mack Avenue, Detroit, MI 48201, USA;
2. Department of Medicinal Chemistry and Molecular Pharmacology, Purdue University, 575 W. Stadium Avenue, West Lafayette, IN 47907, USA;
3. Department of Pharmacology, School of Medicine, Wayne State University, 540 E. Canfield Street, Detroit, MI 48201, USA;
4. Department of Pathology, School of Medicine, Wayne State University, 540 E. Canfield Street, Detroit, MI 48201, USA;
5. Department of Oncology and the Karmanos Cancer Institute, School of Medicine, Wayne State University, 4100 John R Street, Detroit, MI 48201, USA.
Abstract
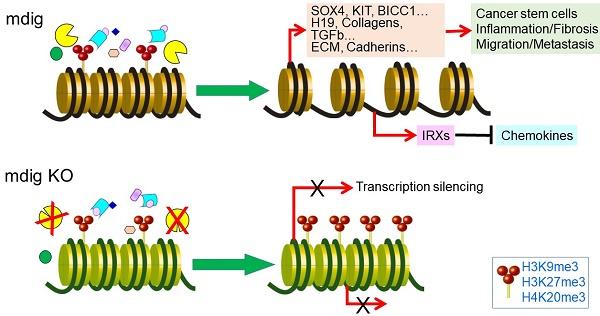
The mineral dust-induced gene (mdig) is overexpressed in a number of human cancers, suggesting critical roles of this gene played on the pathogenesis of cancers. Unlike several other JmjC-domain containing proteins that exhibit histone demethylase activity, it remains enigmatic whether mdig is involved in the demethylation processes of the histone proteins.
Methods: To provide direct evidence suggesting contribution of mdig to the demethylation of histone proteins, we recently examined the histone methylation profiles in human bronchial epithelial cells as well as two cancer cell lines with mdig knockout through CRISPR-Cas9 gene editing.
Results: Global histone methylation analysis revealed a pronounced increase of the repressive histone trimethylation in three different cell types with mdig depletion, including trimethylation of lysines 9 and 27 on histone H3 (H3K9me3, H3K27me3) and trimethylation of lysine 20 of histone H4 (H4K20me3). Importantly, data from both ChIP-seq and RNA-seq suggested that genetic disruption of mdig enriches repressive histone trimethylation and inhibits expression of target genes in the oncogenic pathways of cell growth, stemness of the cells, tissue fibrosis, and cell motility.
Conclusion: Taken together, our study provides the first insight into the molecular effects of mdig as an antagonist for repressive histone methylation markers and suggests that targeting mdig may represent a new area to explore in cancer therapy.
Keywords: Mdig, CRISPR-Cas9, H3K9me3, Cancer stemness, Inflammation.
 Global reach, higher impact
Global reach, higher impact