Impact Factor
Theranostics 2020; 10(2):657-670. doi:10.7150/thno.39132 This issue Cite
Research Paper
A supramolecular protein chaperone for vaccine delivery
1. Tianjin Key Laboratory of Radiation Medicine and Molecular Nuclear Medicine, Institute of Radiation Medicine, Chinese Academy of Medical Sciences & Peking Union Medical College, Tianjin 300192, P.R. China.
2. Key Laboratory of Bioactive Materials, Ministry of Education, College of Life Sciences, State Key Laboratory of Medicinal Chemical Biology, Collaborative Innovation Center of Chemical Science and Engineering, and National Institute of Functional Materials, Nankai University, Tianjin 300071, P. R. China.
3. Analysis Center, Nanjing Medical University, Nanjing, Jiangsu 210029, P. R. China.
4. Lab of Functional and Biomedical Nanomaterials, College of Materials Science and Engineering, Qingdao University of Science and Technology, Qingdao, 266042, P. R. China.
5. Jiangsu Center for the Collaboration and Innovation of Cancer Biotherapy, Cancer Institute, Xuzhou Medical University, Xuzhou, Jiangsu, P. R. China.
Abstract
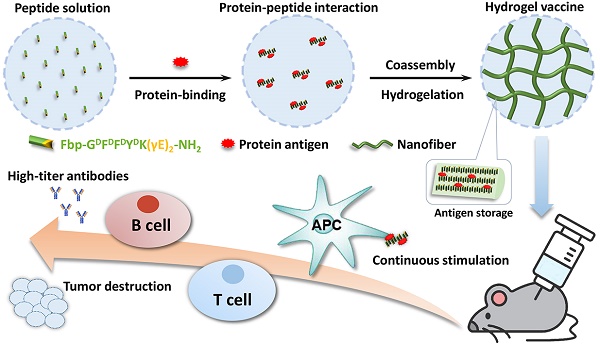
Rationale: Nanomaterials capable of specifically interacting with proteins are very important for protein storage and vaccine delivery. Supramolecular hydrogels based on peptides have emerged as promising vaccine adjuvants because of their good compatibility, ease of antigen incorporation and display, and efficiency in activating immune responses.
Methods: We synthesized a self-assembling peptide (Fbp-GDFDFDYDK(γE)2-NH2, Comp. 1) serving as a supramolecular protein chaperone for protein antigen delivery. The gelation was triggered by simply mixing Comp. 1 and proteins. The vaccine adjuvant potential of Comp. 1 was demonstrated by using two protein antigens, ovalbumin (OVA) and hepatitis B surface antigen (HBsAg).
Results: The peptide derivative Comp. 1 exhibited high protein binding capacity. Upon contacting proteins, Comp. 1 rapidly formed coassembled nanofibers/hydrogels with the proteins, which greatly delayed the release of protein antigens. Our supramolecular protein chaperone significantly stimulated specific antibody titers by assisting protein delivery to antigen-presenting cells, promoting dendritic cell (DC) maturation, prolonging antigen accumulation and retention in the lymph nodes, and eliciting the secretion of cytokines. Most importantly, our supramolecular protein chaperone strongly stimulated the cellular immune response and significantly retarded tumor growth.
Conclusion: Our study demonstrated the great potential of the supramolecular protein chaperone in protein storage and delivery, vaccine production and tumor immunotherapy.
Keywords: Self-assembly, peptide, protein delivery, vaccine adjuvant, immunotherapy
 Global reach, higher impact
Global reach, higher impact