Impact Factor
Theranostics 2020; 10(2):782-796. doi:10.7150/thno.38684 This issue Cite
Research Paper
Theranostic nanobubble encapsulating a plasmon-enhanced upconversion hybrid nanosystem for cancer therapy
1. Department of Chemistry, National Taiwan University, Taipei 106 Taiwan;
2. CAS Key Laboratory of Design and Assembly of Functional Nanostructures, and Fujian Key Laboratory of Nanomaterials, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou, Fujian 350002, China;
3. Genomics Research Center, Academia Sinica, Taipei 115 Taiwan;
4. Department of Biochemistry College of Medicine, Kaohsiung Medical University, Kaohsiung, 807 Taiwan.
5. Department of Mechanical Engineering and Graduate, Institute of Manufacturing Technology, National Taipei University of Technology, Taipei, 106 Taiwan
* These authors contributed equally to this work.
Abstract
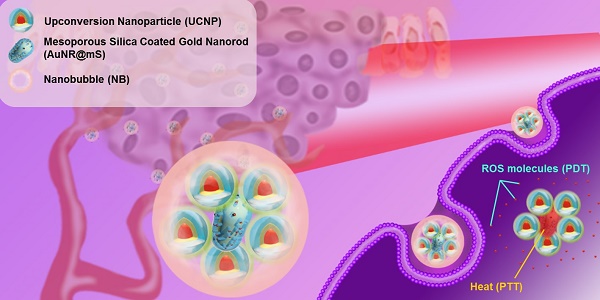
Nanobubble (NB), which simultaneously enhances ultrasound (US) images and access therapeutic platforms, is required for future cancer treatment.
Methods: We designed a theranostic agent for novel cancer treatment by using an NB-encapsulated hybrid nanosystem that can be monitored by US and fluorescent imaging and activated by near-infrared (NIR) light. The nanosystem was transported to the tumor through the enhanced permeability and retention effect. The hybrid nanosystem comprised upconversion nanoparticle (UCNP) and mesoporous silica-coated gold nanorod (AuNR@mS) with the photosensitizer merocyanine 540 to realize dual phototherapy.
Results: With the NIR light-triggered, the luminous intensity of the UCNP was enhanced by doping holmium ion and emitted visible green and red lights at 540 and 660 nm. The high optical density state between the UCNP and AuNR@mS can induce plasmonic enhancement to improve the photothermal and photodynamic effects, resulting in cell death by apoptosis. The nanosystem showed excellent stability to avoid the aggregation of nanoparticles during the treatment. JC-1 dye was used as an indicator of mitochondrial membrane potential to identify the mechanism of cell death. The results of in vitro and in vivo analyses confirmed the curative effect of improved dual phototherapy.
Conclusion: We developed and showed the therapeutic functions of a novel nanosystem with the combination of multiple theranostic nanoplatforms that can be triggered and activated by 808 nm NIR laser and US.
Keywords: near-infrared, plasmonic enhancement, upconversion hybrid nanosystem, dual-model phototherapy, cancer theranostics
 Global reach, higher impact
Global reach, higher impact