13.3
Impact Factor
Theranostics 2020; 10(2):856-866. doi:10.7150/thno.36884 This issue Cite
Research Paper
Therapeutic management of neuro-oncologic patients - potential relevance of CSF liquid biopsy
1. Department of Neurology, LMU Munich, Munich, Germany;
2. Institute of Pathology, LMU Munich, Munich, Germany;
3. German Cancer Consortium (DKTK), Heidelberg, Germany;
4. Department of Medicine III, University Hospital, LMU Munich, Munich, Germany;
5. Comprehensive Cancer Center Munich, University Hospital, LMU Munich, Munich, Germany;
6. Department of Radiation Oncology, University Hospital, LMU Munich, Munich, Germany;
7. Department of Neurosurgery, LMU Munich, Munich, Germany.
Abstract
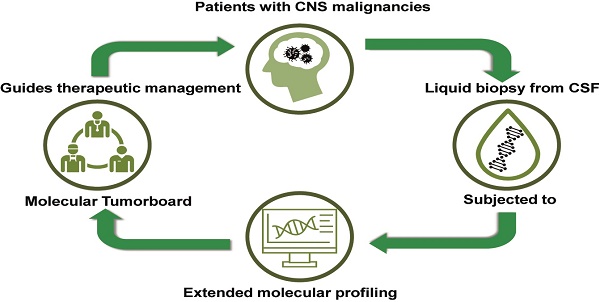
Background: In the era of precision medicine, cancer treatment is increasingly tailored according to tumor-specific genomic alterations. The analysis of tumor-derived circulating nucleic acids in cerebrospinal fluid (CSF) by next generation sequencing (NGS) may facilitate precision medicine in the field of CNS cancer. We therefore evaluated whether NGS from CSF of neuro-oncologic patients reliably detects tumor-specific genomic alterations and whether this may help to guide the management of patients with CNS cancer in clinical practice.
Patient and methods: CSF samples from 27 patients with various primary and secondary CNS malignancies were collected and evaluated by NGS using a targeted, amplicon-based NGS-panel (Oncomine Focus Assay). All cases were discussed within the framework of a molecular tumor board at the Comprehensive Cancer Center Munich.
Results: NGS was technically successful in 23/27 patients (85%). Genomic alterations were detectable in 20/27 patients (74%), 11/27 (40%) of which were potentially actionable. After discussion in the MTB, a change of therapeutic management was recommended in 7/27 (26%) of the cases. However, due to rapid clinical progression, only 4/27 (15%) of the patients were treated according to the recommendation. In a subset of patients (6/27, 22%), a high number of mutations of unknown significance suggestive of a high tumor mutational burden (TMB) were detected.
Conclusions: NGS from cerebrospinal fluid is feasible in routine clinical practice and yields therapeutically relevant alterations in a large subset of patients. Integration of this approach into a precision cancer medicine program might help to improve therapeutic options for patients with CNS cancer.
Keywords: Next Generation Sequencing (NGS), CNS Cancer, CSF, cfDNA, liquid biopsy.
 Global reach, higher impact
Global reach, higher impact