Impact Factor
Theranostics 2020; 10(2):867-879. doi:10.7150/thno.37930 This issue Cite
Research Paper
Red blood cell membrane-enveloped O2 self-supplementing biomimetic nanoparticles for tumor imaging-guided enhanced sonodynamic therapy
1. Britton Chance Center for Biomedical Photonics at Wuhan National Laboratory for Optoelectronics-Hubei Bioinformatics & Molecular Imaging Key Laboratory, Department of Biomedical Engineering, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan 430074, Hubei, P. R. China;
2. Beijing Advanced Innovation Center for Biomedical Engineering, Beijing Advanced Innovation Center for Big Data-Based Precision Medicine, School of Medicine, Beihang University, Beijing, 100083, P. R. China;
3. Key Laboratory of Biomedical Photonics (HUST), Ministry of Education, Huazhong University of Science and Technology, Wuhan 430074, Hubei, P. R. China.
Abstract
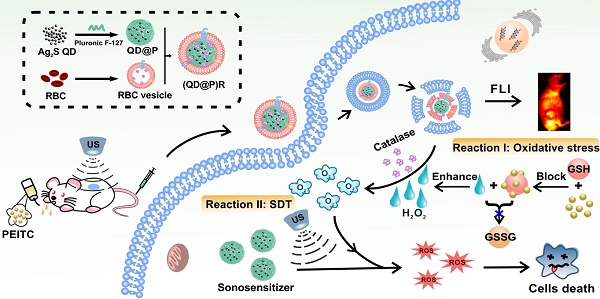
Non-invasive sonodynamic therapy (SDT) was developed because of its advantages of high penetration depth and low side effects; however, tumor hypoxia greatly restricts its therapeutic effect. In this study, we aimed to develop ideal O2 self-supplementing nanoparticles for imaging-guided enhanced sonodynamic therapy of tumors with the adept coalescence of biology with nanotechnology.
Methods: Based on the natural enzyme system of red blood cells (RBC), biomimetic nanoparticles (QD@P)Rs were fabricated by encapsulating Ag2S quantum dots (QD) in RBC vesicle membranes. The anti-tumor drug PEITC was employed to increase the intracellular H2O2 concentration in tumor cells.
Results: In vitro and in vivo experiments demonstrated excellent biocompatibility and prolonged blood circulation of (QD@P)Rs. Following oral administration of PEITC in mice to improve the H2O2 concentration, the enzyme in the nanoprobe catalyzed endogenous H2O2 to increase O2 content and effectively alleviate tumor hypoxia. Triggered by ultrasound under the guidance of fluorescence imaging, (QD@P)Rs generated reactive oxygen species (ROS) to induce tumor cell death, and the increased content of O2 significantly enhanced the effect of SDT.
Conclusion: Ag2S QDs were used, for the first time, as a sonosensitizer in the SDT field. In this study, we integrated the advantages of the natural enzyme system and SDT to develop a novel approach for effective non-invasive treatment of cancer.
Keywords: tumor hypoxia, sonodynamic therapy, red blood cells, nanomedicine, cancer therapy.
 Global reach, higher impact
Global reach, higher impact