Impact Factor
Theranostics 2020; 10(3):998-1015. doi:10.7150/thno.40076 This issue Cite
Research Paper
Activation of JNK and p38 MAPK Mediated by ZDHHC17 Drives Glioblastoma Multiforme Development and Malignant Progression
1. Anhui Province Key Laboratory of Medical Physics and Technology; Center of Medical Physics and Technology, Hefei Institutes of Physical Science, Chinese Academy of Sciences, No. 350, Shushan Hu Road, Hefei, Anhui, 230031, China
2. Department of Molecular Pathology, Hefei Cancer Hospital, Chinese Academy of Sciences, No. 350, Shushan Hu Road, Hefei, Anhui, 230031, China
3. Key Laboratory of the Ministry of Education for Experimental Teratology, Department of Human Anatomy and Histoembryology, School of Basic Medical Sciences, Shandong University, No. 44, Wenhua Xi Road, Jinan, Shandong, 250012, China
4. Department of Hand and Foot Surgery, Second Affiliated Hospital of Shandong University, No.247, Beiyuan Road, Jinan, Shandong, 250033, China
5. University of Science and Technology of China, No. 96, Jin Zhai Road, Hefei, Anhui, 230026, China
6. Department of Radiation Oncology, First Affiliated Hospital, Anhui Medical University, No. 218, Jixi Road, Hefei, Anhui, 230031, China
7. Anhui Key Laboratory for Cellular Dynamics & Chemical Biology, University of Science and Technology of China, Hefei, Anhui, 230026, China
Abstract
Rationale: Glioblastoma multiforme (GBM) almost invariably gain invasive phenotype with limited therapeutic strategy and ill-defined mechanism. By studying the aberrant expression landscape of gliomas, we find significant up-regulation of p-MAPK level in GBM and a potent independent prognostic marker for overall survival. DHHC family was generally expressed in glioma and closely related to the activation of MAPK signaling pathway, but its role and clinical significance in GBM development and malignant progression are yet to be determined.
Method: Bioinformatics analysis, western blotting and immunohistochemistry (IHC) were performed to detect the expression of ZDHHC17 in GBM. The biological function of ZDHHC17 was demonstrated by a series of in vitro and in vivo experiments. Pharmacological treatment, flow cytometry, Transwell migration assay, Co- Immunoprecipitation and GST pulldown were carried out to demonstrate the potential mechanisms of ZDHHC17.
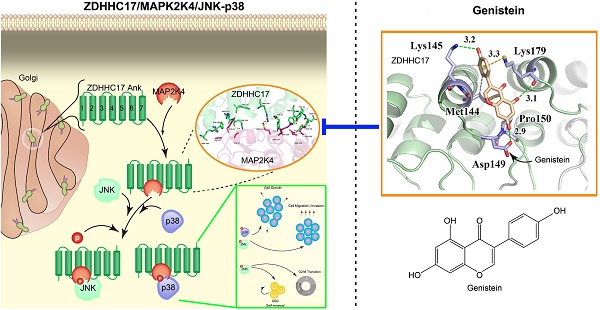
Results: ZDHHC17 is up-regulated and coordinated with MAPK activation in GBM. Mechanistically, ZDHHC17 interacts with MAP2K4 and p38/JNK to build a signaling module for MAPK activation and malignant progression. Notably, the ZDHHC17-MAP2K4-JNK/p38 signaling module contributes to GBM development and malignant progression by promoting GBM cell tumorigenicity and glioma stem cell (GSC) self-renewal. Moreover, we identify a small molecule, genistein, as a specific inhibitor to disrupt ZDHHC17-MAP2K4 complex formation for GBM cell proliferation and GSC self-renewal. Moreover, genistein, identified herein as a lead candidate for ZDHHC17-MAP2K4 inhibition, demonstrated potential therapeutic effect in patients with ZDHHC17-expressing GBM.
Conclusions: Our study identified disruption of a previously unrecognized signaling module as a target strategy for GBM treatment, and provided direct evidence of the efficacy of its inhibition in glioma using a specific inhibitor.
Keywords: glioblastoma multiforme (GBM), ZDHHC17, MAP2K4, JNK/p38, MAPK activation, tumorigenicity, stem cell self-renewal, genistein
 Global reach, higher impact
Global reach, higher impact