Impact Factor
Theranostics 2020; 10(3):1016-1032. doi:10.7150/thno.38537 This issue Cite
Research Paper
PTPN13 induces cell junction stabilization and inhibits mammary tumor invasiveness
1. IRCM, Institut de Recherche en Cancérologie de Montpellier, INSERM U1194, Université de Montpellier, Institut régional du Cancer de Montpellier, Montpellier, F-34298, France.
2. RHEM, BioCampus Montpellier, CNRS, INSERM, University of Montpellier, Montpellier, France.
3. Dynamique des Interactions Membranaires Normales et Pathologiques, CNRS, UMR5235, Montpellier, France.
4. Institute of Functional Genomics, Montpellier, France
5. Department of Pathology, CHU Montpellier, Montpellier, France.
6. Department of Cell Biology, Radboud University Medical Center, Radboud Institute for Molecular Life Sciences, Nijmegen, The Netherlands.
Abstract
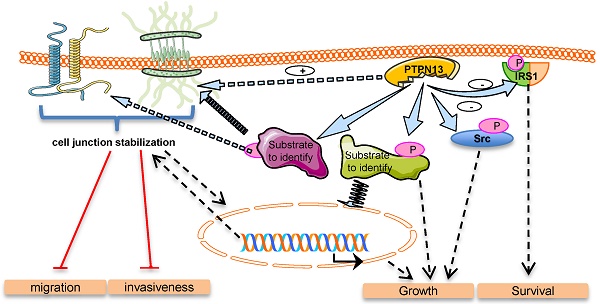
Clinical data suggest that the protein tyrosine phosphatase PTPN13 exerts an anti-oncogenic effect. Its exact role in tumorigenesis remains, however, unclear due to its negative impact on FAS receptor-induced apoptosis.
Methods: We crossed transgenic mice deleted for PTPN13 phosphatase activity with mice that overexpress human HER2 to assess the exact role of PTPN13 in tumor development and aggressiveness. To determine the molecular mechanism underlying the PTPN13 tumor suppressor activity we developed isogenic clones of the aggressive human breast cancer cell line MDA-MB-231 overexpressing either wild type or a catalytically-inactive mutant PTPN13 and subjected these to phosphoproteomic and gene ontology analyses.
We investigated the PTPN13 consequences on cell aggressiveness using wound healing and Boyden chamber assays, on intercellular adhesion using videomicroscopy, cell aggregation assay and immunofluorescence.
Results: The development, growth and invasiveness of breast tumors were strongly increased by deletion of the PTPN13 phosphatase activity in transgenic mice. We observed that PTPN13 phosphatase activity is required to inhibit cell motility and invasion in the MDA-MB-231 cell line overexpressing PTPN13. In vivo, the negative PTPN13 effect on tumor invasiveness was associated with a mesenchymal-to-epithelial transition phenotype in athymic mice xenografted with PTPN13-overexpressing MDA-MB-231 cells, as well as in HER2-overexpressing mice with wild type PTPN13, compared to HER2-overexpressing mice that lack PTPN13 phosphatase activity. Phosphoproteomic and gene ontology analyses indicated a role of PTPN13 in the regulation of intercellular junction-related proteins. Finally, protein localization studies in MDA-MB-231 cells and HER2-overexpressing mice tumors confirmed that PTPN13 stabilizes intercellular adhesion and promotes desmosome formation.
Conclusions: These data provide the first evidence for the negative role of PTPN13 in breast tumor invasiveness and highlight its involvement in cell junction stabilization.
Keywords: Breast cancer, Tyrosine Phosphatase, Transgenic mice, metastases, cell junctions, PTPN13.
 Global reach, higher impact
Global reach, higher impact