Impact Factor
Theranostics 2020; 10(24):10908-10924. doi:10.7150/thno.48264 This issue Cite
Research Paper
Circular RNA circPPM1F modulates M1 macrophage activation and pancreatic islet inflammation in type 1 diabetes mellitus
1. Institute of Pediatrics, Children's Hospital of Fudan University, and the Shanghai Key Laboratory of Medical Epigenetics, International Co-laboratory of Medical Epigenetics and Metabolism, Ministry of Science and Technology, Institutes of Biomedical Sciences, Fudan University, Shanghai 200032, China.
2. National Health Commission (NHC) Key Laboratory of Neonatal Diseases, Fudan University, Shanghai, China.
3. Department of Pediatric Endocrinology and Inherited Metabolic Diseases, Children's Hospital of Fudan University, Shanghai, 201102, China.
#These authors contributed equally to this work.
Abstract
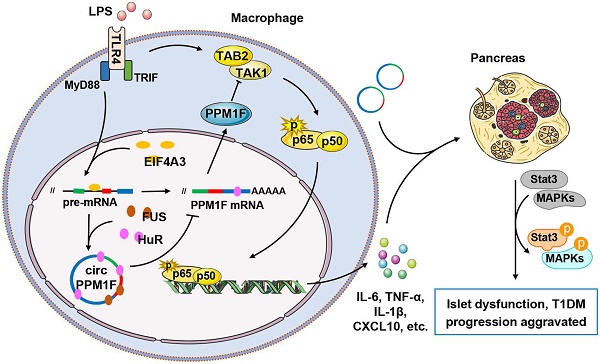
Rationale: Macrophages play critical roles in the pathogenesis of type 1 diabetes mellitus (T1DM). Circular RNAs (circRNAs) are a novel class of endogenous RNAs with covalently closed loop structures, implicated in various disease processes. However, their impact on macrophage activation and T1DM pathogenesis remains elusive.
Methods: circRNA expression profiles of peripheral blood mononuclear cells (PBMCs) from T1DM children were determined by whole transcriptome microarray. Bioinformatics, quantitative real-time PCR, Western blot, RNA immunoprecipitation (RIP), cell co-culture, cell proliferation, and cell apoptosis assays were performed to investigate the expression, function, and regulatory mechanisms of circPPM1F in vitro. The regulatory role of circPPM1F in vivo was evaluated in the streptozocin-induced diabetic mouse model.
Results: We identified 27 upregulated and 31 downregulated differentially expressed circRNAs in T1DM patients. circPPM1F, a circRNA with unknown function, was dominantly expressed in monocytes and significantly upregulated in T1DM patients. Functionally, circPPM1F promoted lipopolysaccharide (LPS)-induced M1 macrophage activation via enhancement of the NF-κB signaling pathway. Mechanistically, circPPM1F competitively interacted with HuR to impair the translation of protein phosphatase, Mg2+/Mn2+ dependent 1F (PPM1F), thus alleviating the inhibitory effect of PPM1F on the NF-κB pathway. Moreover, eukaryotic initiation factor 4A-III (EIF4A3) and fused in sarcoma (FUS) coordinately regulated circPPM1F expression during M1 macrophage activation. In addition, circPPM1F could exacerbate pancreas injury in the streptozocin-induced diabetic mice by activation of M1 macrophages in vivo.
Conclusions: circPPM1F is a novel positive regulator of M1 macrophage activation through the circPPM1F-HuR-PPM1F-NF-κB axis. Overexpression of circPPM1F could promote pancreatic islet injury by enhancing M1 macrophage activation and circPPM1F may serve as a novel potential therapeutic target for T1DM in children.
Keywords: Circular RNA, Type 1 diabetes mellitus, Macrophage activation, RNA-binding protein, Islet injury
 Global reach, higher impact
Global reach, higher impact