Impact Factor
Theranostics 2020; 10(24):11092-11109. doi:10.7150/thno.44997 This issue Cite
Research Paper
Truncated HDAC9 identified by integrated genome-wide screen as the key modulator for paclitaxel resistance in triple-negative breast cancer
1. Department of Breast Surgery, Key Laboratory of Breast Cancer in Shanghai, Fudan University Shanghai Cancer Center, Fudan University, Shanghai, China.
2. Department of Oncology, Shanghai Medical College, Fudan University, Shanghai, China.
3. Precision Cancer Medicine Center, Fudan University Shanghai Cancer Center, Shanghai, China.
4. Laboratory of Systems Biology, Shanghai Advanced Research Institute, Chinese Academy of Sciences, 200031 Shanghai, China.
*These authors contributed equally to this work.
Abstract
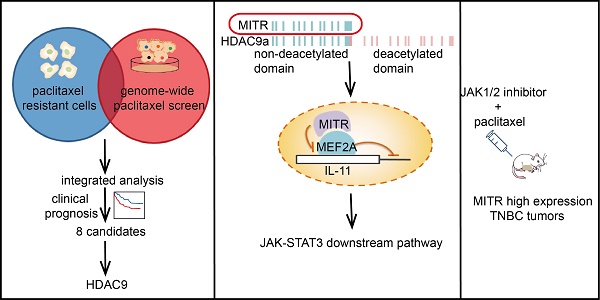
Rationale: Paclitaxel resistance is a major concern when treating triple-negative breast cancer (TNBC) patients. We aimed to identify candidates causing paclitaxel resistance and explore their significance in TNBC therapeutics.
Methods: A genome-wide CRISPR screening, integrated with transcriptome analyses, was performed to identify candidates involved in paclitaxel-resistant TNBCs. Cell proliferation, cytotoxicity, immunofluorescent staining, and xenograft assays were conducted to verify the phenotypes of paclitaxel resistance induced by candidate genes, both in vitro and in vivo. RNA sequencing, Western blotting, and chromatin immunoprecipitation assays were used to explore the underlying mechanisms.
Results: MEF2-interacting transcriptional repressor (MITR), the truncated isoform of histone deacetylase 9 (HDAC9) lacking the deacetylation domain, was enriched in paclitaxel-resistant cells. Elevated MITR expression resulted in increased interleukin-11 (IL11) expression and activation of downstream JAK/STAT3 signaling. Mechanistically, MITR counteracted MEF2A-induced transcriptional suppression of IL11, ultimately causing paclitaxel resistance. By contrast, pharmacological inhibition of JAK1/2 by ruxolitinib reversed paclitaxel resistance both in vitro and in vivo.
Conclusion: Our in vitro and in vivo genetic and cellular analyses elucidated the pivotal role of MITR/MEF2A/IL11 axis in paclitaxel resistance and provided a novel therapeutic strategy for TNBC patients to overcome poor chemotherapy responses.
Keywords: breast cancer, paclitaxel resistance, CRISPR screen, HDAC9, MITR
 Global reach, higher impact
Global reach, higher impact