Impact Factor
Theranostics 2020; 10(24):11159-11177. doi:10.7150/thno.44389 This issue Cite
Research Paper
Rbm24 modulates adult skeletal muscle regeneration via regulation of alternative splicing
1. Institute of Stem Cell and Regenerative Medicine, School of Medicine, Xiamen University, Xiamen 361100, China.
2. Shenzhen Research Institute of Xiamen University, Guangdong Province, 518000, P.R. China.
3. Fujian Heze Biological Technology Co., Ltd, Xiamen, Fujian, China.
4. Eye Institute of Xiamen University, Fujian Provincial Key Laboratory of Ophthalmology and Visual Science, Xiamen, Fujian, China.
Abstract
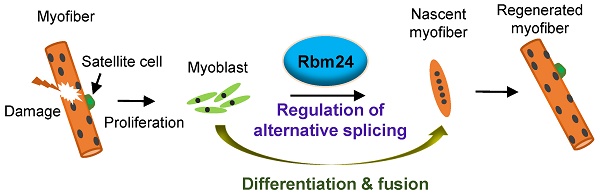
Rationale: The adult skeletal muscle can self-repair efficiently following mechanical or pathological damage due to its remarkable regenerative capacity. However, regulatory mechanisms underlying muscle regeneration are complicated and have not been fully elucidated. Alternative splicing (AS) is a major mechanism responsible for post-transcriptional regulation. Many aberrant AS events have been identified in patients with muscular dystrophy which is accompanied by abnormal muscle regeneration. However, little is known about the correlation between AS and muscle regeneration. It has been reported that RNA binding motif protein 24 (Rbm24), a tissue-specific splicing factor, is involved in embryo myogenesis while the role of Rbm24 in adult myogenesis (also called muscle regeneration) is poorly understood.
Methods: To investigate the role of Rbm24 in adult skeletal muscle, we generated Rbm24 conditional knockout mice and satellite cell-specific knockout mice. Furthermore, a cardiotoxin (CTX)-induced injury model was utilized to assess the effects of Rbm24 on skeletal muscle regeneration. Genome-wide RNA-Seq was performed to identify the changes in AS following loss of Rbm24.
Results: Rbm24 knockout mice displayed abnormal regeneration 4 months after tamoxifen treatment. Using RNA-Seq, we found that Rbm24 regulated a complex network of AS events involved in multiple biological processes, including myogenesis, muscle regeneration and muscle hypertrophy. Moreover, using a CTX-induced injury model, we showed that loss of Rbm24 in skeletal muscle resulted in myogenic fusion and differentiation defects and significantly delayed muscle regeneration. Furthermore, satellite cell-specific Rbm24 knockout mice recapitulated the defects in regeneration seen in the global Rbm24 knockout mice. Importantly, we demonstrated that Rbm24 regulated AS of Mef2d, Naca, Rock2 and Lrrfip1 which are essential for myogenic differentiation and muscle regeneration.
Conclusions: The present study demonstrated that Rbm24 regulates dynamic changes in AS and is essential for adult skeletal muscle regeneration.
Keywords: RNA binding motif protein 24, muscle injury, muscle regeneration, alternative splicing, myogenic differentiation
 Global reach, higher impact
Global reach, higher impact