Impact Factor
Theranostics 2020; 10(3):1151-1165. doi:10.7150/thno.38327 This issue Cite
Research Paper
Chimeric peptide supramolecular nanoparticles for plectin-1 targeted miRNA-9 delivery in pancreatic cancer
1. Cancer Institute of Integrated Traditional Chinese and Western Medicine, Zhejiang Academy of Traditional Chinese Medicine, Tongde Hospital of Zhejiang province, Hangzhou 310012, China
2. Department of Genetics, Institute of Genetics, Institute of Cell Biology, Zhejiang University School of Medicine, Hangzhou 310058, China
*These authors contributed equally to this work
Abstract
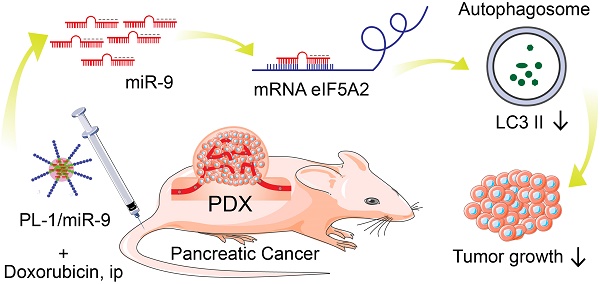
Pancreatic ductal adenocarcinoma (PDAC) is a highly lethal disease with poor prognosis. Insights into the roles of MicroRNAs (miRNAs) in diseases, particularly in cancer, have made miRNAs attractive tools and targets for novel therapeutic approaches.
Methods: Here, we employed a novel chimeric peptide supramolecular nanoparticle delivery system for plectin-1 (PL-1)-targeted PDAC-specific miR-9 delivery in vitro and in pancreatic cancer patient-derived xenograft (PDX) model. RT-PCR and immunohistochemistry (IHC) were conducted to detect the expression pattern of eIF5A2. mRFP-GFP-LC3 fluorescence microscopy and Western blot were carried out to determine autophagy. Luciferase reporter assays were performed to elucidate the regulatory role of miR-9/eIF5A2 axis.
Results: PL-1/miR-9 nanocomplexes dramatically improve the anticancer effect of doxorubicin through downregulating eIF5A2 expression to inhibit autophagy and induce apoptosis in PDAC therapy in vivo. Mechanistically, miR-9 directly targets the eIF5A2 transcript by binding to its 3'-untranslated region (3'-UTR) to reduce the expression levels and the secreted protein of eIF5A2 in PDAC cells.
Conclusion: PL-1/miR-9 nanoparticles can be used as a novel promising anti-cancer strategy with tumor targeting and miR-9/eIF5A2 may serve as a new potential therapeutic target for future synergic therapy against human PDAC.
Keywords: PDAC, miR-9, nanoparticle, eIF5A2, autophagy
 Global reach, higher impact
Global reach, higher impact