13.3
Impact Factor
Theranostics 2020; 10(3):1181-1196. doi:10.7150/thno.38989 This issue Cite
Research Paper
Targeted destruction of cancer stem cells using multifunctional magnetic nanoparticles that enable combined hyperthermia and chemotherapy
1. Department of Biomedical Sciences, City University of Hong Kong, Hong Kong, China.
2. Key Laboratory of Biochip Technology, Biotech and Health Centre, Shenzhen Research Institute of City University of Hong Kong, Shenzhen 518057, China.
3. College of Chemistry and Environmental Science, Key Laboratory of Medicinal Chemistry and Molecular Diagnosis of the Ministry of Education, Hebei University, Baoding 071002, China.
4. Department of Thoracic Surgery, Shenzhen People's Hospital, the Second Clinical Medical College of Jinan University, Shenzhen 510000, China.
5. Department of Clinical Oncology, Queen Elizabeth Hospital, Hong Kong, China.
6. Department of Oncology, Peking University Shenzhen Hospital, Shenzhen 518036, China.
✉ Corresponding author: Mengsu Yang. Tel.: (852) 3442-7797; Fax: (852) 3442-0552; Email: bhmyang@cityu.edu.hk
Abstract
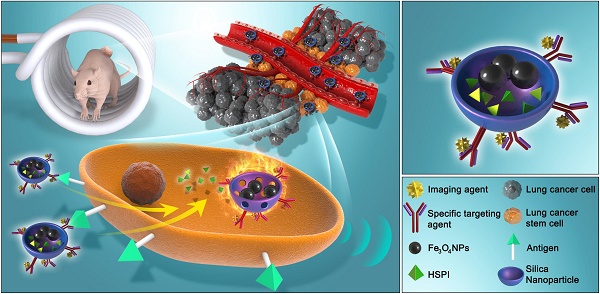
Cancer stem cells (CSCs) have been implicated in cancer recurrence and therapy resistance. Therefore, a CSC-targeted therapy that disrupts the maintenance and survival of CSCs may offer an effective approach in killing tumor cells in primary tumors and preventing the metastasis caused by CSCs. Nanoparticles (NPs)-based thermotherapy and/or chemotherapy are promising therapeutic methods for cancer treatment.
Methods: A silica-based multifunctional NP system was present, which encapsulated a chemotherapeutic agent and magnetic cores and coated with a specific antibody against the lung CSCs. The efficacy of this novel therapeutic strategy was systematically studied both in vitro and in vivo by simultaneous activating the combined thermotherapy and chemotherapy via CSC-targeted NPs.
Results: These NPs were systematically administered and activated for targeted chemotherapy and thermotherapy by using an externally applied alternating magnetic field (AMF). The antibody-modified NPs targeted to lung CSCs with enhanced cellular uptake in vitro and extended accumulation in tumor in vivo. Up to 98% of lung CSCs was killed in vitro with 30-min application of AMF, due to the combined effects of hyperthermia and chemotherapeutic drug treatment. In in vivo models, this combined therapy significantly suppressed tumor growth and metastasis in lung CSC xenograft-bearing mice, with minimal side effects and adverse effects.
Conclusion: With good biocompatibility and targeting capability, the nanodrug delivery system may offer a promising clinical platform for the combined thermotherapy and chemotherapy. This work demonstrated the feasibility of developing multifunctional nanomedicine targeting CSCs for effective cancer treatment.
Keywords: Lung cancer stem cell, Multifunctional nanoparticle, Alternating magnetic field, Thermotherapy and chemotherapy
 Global reach, higher impact
Global reach, higher impact