Impact Factor
Theranostics 2020; 10(3):1213-1229. doi:10.7150/thno.38900 This issue Cite
Research Paper
Proteomic analysis of intracellular protein corona of nanoparticles elucidates nano-trafficking network and nano-bio interactions
1. Beijing Key Laboratory of Molecular Pharmaceutics and New Drug Delivery Systems, School of Pharmaceutical Sciences, Peking University, Beijing, 100191, China.
2. State Key Laboratory of Natural and Biomimetic Drugs, Peking University, Beijing, 100191, China.
3. School of Food and Biological Engineering, Shaanxi University of Science and Technology, Xi'an, 710021, China.
4. School of Pharmaceutical Science, Guangzhou Medical University, Guangzhou, 511436, China.
Abstract
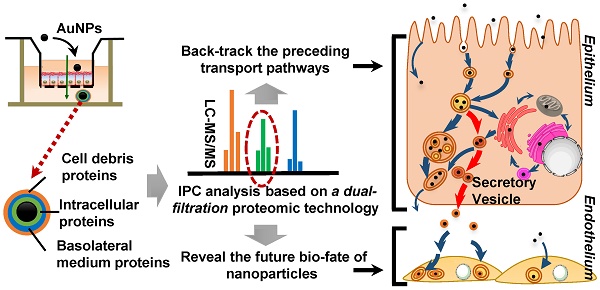
The merits of nanomedicines are significantly impacted by the surrounding biological environment. Similar to the protein corona generated on the surface of nanoparticles in the circulation system, the intracellular protein corona (IPC) might be formed on nanoparticles when transported inside the cells. However, little is known currently about the formation of IPC and its possible biological influence.
Methods: Caco-2 cells, a classical epithelial cell line, were cultured in Transwell plates to form a monolayer. Gold nanoparticles (AuNPs) were prepared as the model nanomedicine due to their excellent stability. Here we focused on identifying IPC formed on the surface of AuNPs during cell transport. The nanoparticles in the basolateral side of the Caco-2 monolayer were collected and analyzed by multiple techniques to verify IPC formation. High-performance liquid chromatography-tandem mass spectrometry (LC-MS/MS)-based proteomics was utilized to analyze the composition of IPC proteins. In particular, we established a dual-filtration strategy to exclude various interference in IPC identification. Based on the subcellular localization of specific IPC proteins, we elicited the nano-trafficking network of AuNPs. The transport pathways of AuNPs identified by proteomic analysis were also verified by various conventional technologies. Finally, we explored the influence of IPC on the uptake and stress response of endothelium.
Results: The existence of IPC was demonstrated on the surface of AuNPs, in which 227 proteins were identified. Among them, 40 proteins were finally ascertained as the specific IPC proteins. The subcellular location analysis indicated that these “specific” IPC proteins could back-track the transport pathways of nanoparticles in the epithelial cell monolayer. According to the subcellular distribution of IPC proteins and co-localization, we discovered a new pathway of nanoparticles from endosomes to secretory vesicles which was dominant during the transcytosis. After employing conventional imageology and pharmacology strategies to verify the result of proteomic analysis, we mapped a comprehensive intracellular transport network. Our study also revealed the merits of IPC analysis, which could readily elucidate the molecular mechanisms of transcytosis. Besides, the IPC proteins increased the uptake and stress response of endothelium, which was likely mediated by extracellular matrix and mitochondrion-related IPC proteins.
Conclusion: The comprehensive proteomic analysis of IPC enabled tracing of transport pathways in epithelial cells as well as revealing the biological impact of nanoparticles on endothelium.
Keywords: intracellular protein corona, quantitative proteomics, nanoparticles, transcytosis, nano-bio interaction.
 Global reach, higher impact
Global reach, higher impact