Impact Factor
Theranostics 2020; 10(3):1433-1453. doi:10.7150/thno.38507 This issue Cite
Research Paper
MicroRNA-204-5p is a tumor suppressor and potential therapeutic target in head and neck squamous cell carcinoma
1. Department of Oral and Maxillofacial Surgery, Guanghua School of Stomatology, Hospital of Stomatology, Sun Yat-sen University, Guangzhou 510055, China.
2. Guangdong Provincial Key Laboratory of Stomatology, Sun Yat-sen University, Guangzhou 510080, China.
3. Department of Oral Pathology, Guanghua School of Stomatology, Hospital of Stomatology, Sun Yat-sen University, Guangzhou 510055,China.
4. Department of Operative Dentistry and Endodontics, Guanghua School of Stomatology, Hospital of Stomatology, Sun Yat-sen University, Guangzhou 510055, China.
5. School of Life Science, Sun Yat-sen University, Guangzhou 510275, China.
6. South China Institute of Biomedine, Guangzhou 510535, China.
7. State Key Laboratory of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, Guangzhou 510060, China.
8. Department of Medicinal Chemistry, School of Pharmacy, and the Massey Cancer Center, Virginia Commonwealth University, Richmond, Virginia 23298-0540, United States.
9. Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University, Richmond, Virginia 23298-0540, Unites States.
*These authors contributed equally to this work.
Abstract
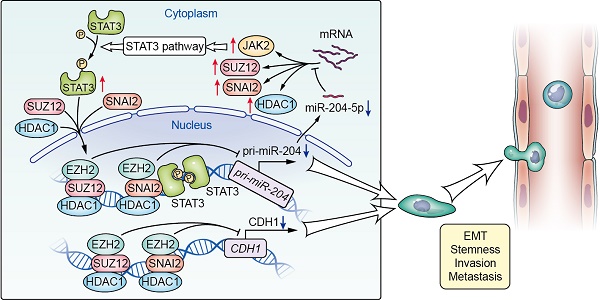
Elucidation of the molecular mechanisms governing aggressiveness of HNSCC may provide clinical therapeutic strategies for patients. In this study, a novel hub miR-204-5p functioning as tumor suppressor has been identified and explored in HNSCC.
Methods: A novel hub miR-204-5p was identified based on miRNA microarray, bioinformatics analysis and validated in different HNSCC patient cohorts. The functional role of miR-204-5p and its downstream and upstream regulatory machinery were investigated by gain-of-function and loss-of-function assays in vitro and in vivo. Interactions among miR-204-5p and SNAI2/SUZ12/HDAC1/STAT3 complex were examined by a series of molecular biology experiments. Then, the clinical relevance of miR-204-5p and its targets were evaluated in HNSCC samples. HNSCC patient-derived xenograft (PDX) model was used to assess the therapeutic value of miR-204-5p.
Results: We reveal that miR-204-5p as a tumor suppressor is commonly repressed in HNSCC, which can inhibit tumor growth, metastasis and stemness. Mechanically, miR-204-5p suppresses epithelial-mesenchymal transition (EMT) and STAT3 signaling by targeting SNAI2, SUZ12, HDAC1 and JAK2. Among these targets, we further showed that SNAI2, SUZ12, and HDAC1 form a repressive complex on CDH1 promoter to maintain EMT in HNSCC. In turn, the SNAI2/SUZ12/HDAC1 complex interacts with STAT3 on miR-204-5p-regulatory regions to suppress the transcription of miR-204-5p. Moreover, we also show that decrease of miR-204-5p indicates a poor prognosis in HNSCC patients and administration of agomiR-204-5p inhibits tumor growth and metastasis in HNSCC PDX models.
Conclusion: miR-204-5p-SNAI2/SUZ12/HDAC1/STAT3 regulatory circuit has a critical role in maintaining aggressiveness of HNSCC, suggesting that miR-204-5p might serve as a promising therapeutic target for clinical intervention.
Keywords: miR-204-5p, tumor suppressor, metastasis, tumorigencity, head and neck squamous cell carcinoma
 Global reach, higher impact
Global reach, higher impact