Impact Factor
Theranostics 2020; 10(4):1849-1860. doi:10.7150/thno.40869 This issue Cite
Research Paper
Label-free whole-colony imaging and metabolic analysis of metastatic pancreatic cancer by an autoregulating flexible optical system
Key Laboratory of Optoelectronic Devices and Systems of Guangdong Province & Ministry of Education, College of Physics and Optoelectronic Engineering, Shenzhen University, Shenzhen, China.
*These authors contributed equally.
Abstract
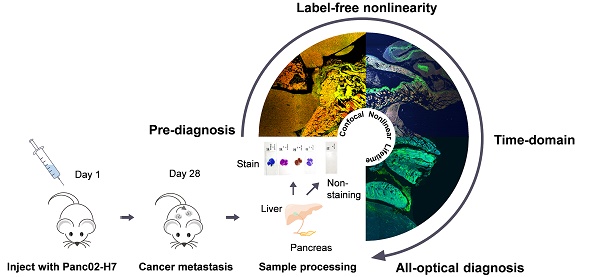
Cancer metastasis is a Gordian knot for tumor diagnosis and therapy. Many studies have demonstrated that metastatic processes are inevitably affected by the tumor microenvironment. Histopathology is used universally as the gold standard for cancer diagnosis despite the lengthy preparation process and invasiveness.
Methods: Here, we introduced a supercontinuum and super-wide-tuning integrated multimodal platform, which combines the confocal, nonlinear and fluorescence lifetime microscopy with autoregulations, for label-free evaluation of fresh tissue and pathological sections. Based on various automated tunable lasers, synchronized and self-adjusting components and eight fast switching detection channels, the system features fast, large-field and subcellular-scale imaging of exogenous and endogenous fluorophores, nonlinear coherent scattering and lifetime contrast.
Results: With such an integrated multi-dimensional system, we searched the metastatic region by two-photon and three-photon excited autofluorescence, analyzed the cancer invasion by second harmonic generation and revealed the affected cellular metabolism by phasor-lifetime. We demonstrated the flexible measurement of multiple nonlinear modalities at NIR I and II excitation with a pre-compensation for group delay dispersion of ~7,000 fs2 and low power of <40 mW, and of dual autofluorescence lifetime decays for phasor approach to decompose cancer-associated and disassociated components. This significantly revealed the metastatic and metabolic optical signatures of the whole colony of pancreatic cancers.
Conclusion: The synergistic effect of the system demonstrates the great potential to translate this technique into routine clinical applications, particularly for large-scale and quantitative studies of metastatic colonization.
Keywords: metastatic cancer, label-free imaging, nonlinear optical imaging, autofluorescence lifetime, phasor approach
 Global reach, higher impact
Global reach, higher impact