Impact Factor
Theranostics 2020; 10(5):2095-2114. doi:10.7150/thno.30736 This issue Cite
Research Paper
Anti-CDCP1 immuno-conjugates for detection and inhibition of ovarian cancer
1. Mater Research Institute - The University of Queensland, Translational Research Institute, Woolloongabba, QLD 4102, Australia.
2. Mater Ovarian Cancer Research Collaborative, Mater Adult Hospital, South Brisbane, QLD 4101, Australia.
3. Australian Institute for Bioengineering and Nanotechnology, The University of Queensland, St Lucia, Australia.
4. Commonwealth Scientific and Industrial Research Organisation, Probing Biosystems Future Science Platform, Herston, QLD 4029, Australia.
5. Biomedicine Discovery Institute and Department of Biochemistry and Molecular Biology, Monash University, Melbourne, VIC 3800, Australia.
6. Preclinical Imaging Facility, Translational Research Institute, Woolloongabba, QLD 4102, Australia.
7. Centre for Clinical Research, University of Queensland, Herston, Qld 4029, Australia.
8. Institute of Health and Biomedical Innovation, Queensland University of Technology, Woolloongabba, QLD 4102, Australia.
9. Department of Medical Oncology, Mayo Clinic, Rochester, MN, USA.
10. Merck Research Laboratories, Rahway, NJ, USA.
11. Mater Health Services, South Brisbane, QLD 4101, Australia.
Abstract
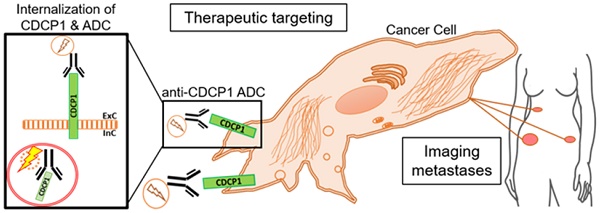
CUB-domain containing protein 1 (CDCP1) is a cancer associated cell surface protein that amplifies pro-tumorigenic signalling by other receptors including EGFR and HER2. Its potential as a cancer target is supported by studies showing that anti-CDCP1 antibodies inhibit cell migration and survival in vitro, and tumor growth and metastasis in vivo. Here we characterize two anti-CDCP1 antibodies, focusing on immuno-conjugates of one of these as a tool to detect and inhibit ovarian cancer.
Methods: A panel of ovarian cancer cell lines was examined for cell surface expression of CDCP1 and loss of expression induced by anti-CDCP1 antibodies 10D7 and 41-2 using flow cytometry and Western blot analysis. Surface plasmon resonance analysis and examination of truncation mutants was used to analyse the binding properties of the antibodies for CDCP1. Live-cell spinning-disk confocal microscopy of GFP-tagged CDCP1 was used to track internalization and intracellular trafficking of CDCP1/antibody complexes. In vivo, zirconium 89-labelled 10D7 was detected by positron-emission tomography imaging, of an ovarian cancer patient-derived xenograft grown intraperitoneally in mice. The efficacy of cytotoxin-conjugated 10D7 was examined against ovarian cancer cells in vitro and in vivo.
Results: Our data indicate that each antibody binds with high affinity to the extracellular domain of CDCP1 causing rapid internalization of the receptor/antibody complex and degradation of CDCP1 via processes mediated by the kinase Src. Highlighting the potential clinical utility of CDCP1, positron-emission tomography imaging, using zirconium 89-labelled 10D7, was able to detect subcutaneous and intraperitoneal xenograft ovarian cancers in mice, including small (diameter <3 mm) tumor deposits of an ovarian cancer patient-derived xenograft grown intraperitoneally in mice. Furthermore, cytotoxin-conjugated 10D7 was effective at inhibiting growth of CDCP1-expressing ovarian cancer cells in vitro and in vivo.
Conclusions: These data demonstrate that CDCP1 internalizing antibodies have potential for killing and detection of CDCP1 expressing ovarian cancer cells.
Keywords: CDCP1, ovarian cancer, immuno-conjugate, antibody
 Global reach, higher impact
Global reach, higher impact