Impact Factor
Theranostics 2020; 10(5):2201-2214. doi:10.7150/thno.39621 This issue Cite
Research Paper
Self-activated in vivo therapeutic cascade of erythrocyte membrane-cloaked iron-mineralized enzymes
1. Department of Orthopedics, The First Affiliated Hospital of Jinan University, Guangzhou 510632, China.
2. Key Laboratory of Biomaterials of Guangdong Higher Education Institutes, Guangdong Provincial Engineering and Technological Research Center for Drug Carrier Development, Department of Biomedical Engineering, Jinan University, Guangzhou 510632, China.
3. Institute of Life and Health Engineering, Key Laboratory of Functional Protein Research of Guangdong Higher Education Institutes, Jinan University, Guangzhou 510632, China.
Abstract
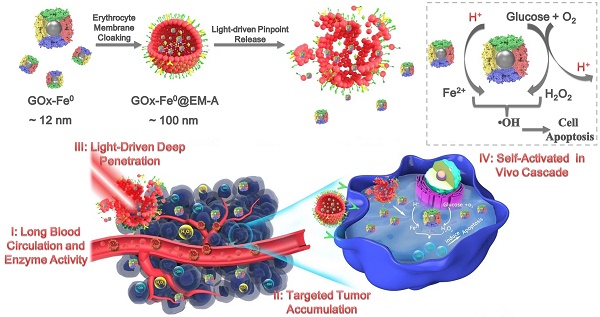
Biomineralization of enzymes for in vivo diagnosis and treatment of diseases remain a considerable challenge, due to their severe reaction conditions and complicated physiological environment. Herein, we reported a biomimetic enzyme cascade delivery nanosystem, tumor-targeted erythrocyte membrane (EM)-cloaked iron-mineralized glucose oxidases (GOx-Fe0@EM-A) for enhancing anticancer efficacy by self-activated in vivo cascade to generate sufficient high toxic •OH at tumor site.
Methods: An ultra-small Fe0 nanoparticle (Fe0NP) was anchored in the inner cavity of glucose oxidase (GOx) to form iron-mineralized glucose oxidase (GOx-Fe0) as a potential tumor therapeutic nanocatalyst. Moreover, erythrocyte membrane cloaking delivery of GOx-Fe0 in vivo was designed to effectively accumulate ultra-small GOx-Fe0 at tumor site.
Results: GOx-Fe0@EM-A had satisfactory biocompatibility and light-trigged release efficiency. Erythrocyte membrane cloaking of GOx-Fe0@EM-A not only prolongs blood circulation but also protects in vivo enzyme activity of GOx-Fe0; Tumor targeting of GOx-Fe0@EM-A endowed preferential accumulation at tumor site. After NIR light irradiation at tumor site, erythrocyte membrane of GOx-Fe0@EM-A was ruptured to achieve light-driven release and tumor deep penetration of ultra-small nanosize GOx-Fe0 by the photothermal effect of ICG. Then, GOx-Fe0 occurred self-activated in vivo cascade to effectively eradicate tumor by producing the highly cumulative and deeply penetrating •OH at tumor site.
Conclusion: Tumor-targeted erythrocyte membrane-cloaked iron-mineralized glucose oxidase (GOx-Fe0@EM-A) exhibits a promising strategy for striking antitumor efficacy by light-driven tumor deep penetration and self-activated therapeutic cascade.
Keywords: enzyme biohybrid, light-driven pinpoint release, self-activated therapeutic cascade, tumor-enhanced penetration, erythrocyte membrane cloaking
 Global reach, higher impact
Global reach, higher impact