13.3
Impact Factor
Theranostics 2020; 10(5):2327-2341. doi:10.7150/thno.39740 This issue Cite
Research Paper
CCBE1 promotes tumor lymphangiogenesis and is negatively regulated by TGFβ signaling in colorectal cancer
1. Department of Colorectal and Anal Surgery, Xinhua Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200092, China
2. State Key Laboratory of Genetic Engineering and Collaborative Innovation Center for Genetics and Development, School of Life Sciences and Zhongshan Hospital, Fudan University, Shanghai 200438, China
3. Suzhou Institute of Systems Medicine, Center for Systems Medicine Research, Chinese Academy of Medical Sciences, Suzhou, Jiangsu 215123, China
4. Shanghai Colorectal Cancer Research Center, Shanghai 200092, China
5. Department of anesthesiology, Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, China
6. The First Department of Gastrointestinal Surgery, Henan Provincial People's Hospital, People's Hospital of Zhengzhou University, People's Hospital of Henan University, Zhengzhou, Henan 450003, China.
*These authors contribute equally.
Abstract
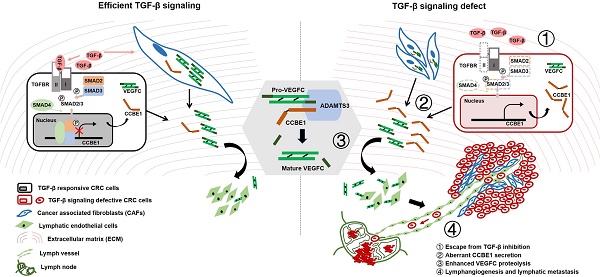
Collagen and calcium-binding EGF domain-1 (CCBE1) is essential for lymphatic vascular development as it promotes vascular endothelial growth factor C (VEGFC) proteolysis. A recent study reported that CCBE1 was overexpressed in epithelial colorectal cancer (CRC) cells; however, the role of CCBE1 in tumor lymphangiogenesis and the mechanism underlying dysregulated CCBE1 expression in CRC remain undefined.
Methods: The role of CCBE1 in tumor lymphangiogenesis and lymphatic metastasis was investigated using human lymphatic endothelial cells (HLECs) model in vitro, and a hindfoot lymphatic metastasis model in vivo. Immunochemistry analysis was performed to assess CCBE1 expression, prognostic value and correlation with clinicopathological characteristics in CRC. The biochemical function and transcriptional regulatory mechanism of CCBE1 were explored by western blot, qPCR, and chromatin immunoprecipitation.
Results: Cancer cell-derived CCBE1 enhances VEGFC proteolysis in vitro, facilitates tube formation and migration of HLECs in vitro, and promotes tumor lymphangiogenesis and lymphatic metastasis in vivo. In addition to CRC cells, tumor stroma within CRC tissue shows high CCBE1 expression, which is associated with high lymphatic vessel density, increased lymph node metastasis and poor prognosis. Cancer-associated fibroblasts (CAFs) express and secret CCBE1, thereby contributing to VEGFC maturation and tumor lymphangiogenesis in CRC. Transforming growth factor beta (TGF-β) downregulates the transcription and lymphangiogenic function of CCBE1 in CAFs and CRC cells through direct binding of SMADs to CCBE1 gene locus. Inactivation of the TGF-β pathway correlates with increased CCBE1 expression in CRC.
Conclusion: Our results demonstrate the protumorigenic role of CCBE1 in promoting lymphangiogenesis and lymphatic metastasis in CRC, revealing a new mechanism by which loss of TGF-β signaling promotes CRC metastasis.
Keywords: CCBE1, colorectal cancer, tumor lymphangiogenesis, lymphatic metastasis, TGF-β
 Global reach, higher impact
Global reach, higher impact