Impact Factor
Theranostics 2020; 10(5):2436-2452. doi:10.7150/thno.37949 This issue Cite
Research Paper
Acute cellular and vascular responses to photodynamic therapy using EGFR-targeted nanobody-photosensitizer conjugates studied with intravital optical imaging and magnetic resonance imaging
1. Center for Optical Diagnostics and Therapy, Dept. of Otolaryngology and Head & Neck Surgery, Erasmus MC Cancer Institute, Rotterdam, The Netherlands.
2. Cell Biology Division, Dept. of Biology, Faculty of Science, Utrecht University, Utrecht, The Netherlands.
3. Biomedical NMR, Biomedical Engineering, Eindhoven University of Technology, Eindhoven, The Netherlands.
4. Division of Optical Molecular Imaging, Dept. of Radiology, Leiden University Medical Center, Leiden, The Netherlands.
5. Amsterdam University Medical Centers, University of Amsterdam, Dept. of Biomedical Engineering and Physics, The Netherlands.
6. Dept. of Pathology, University Medical Centre Utrecht, Utrecht, The Netherlands.
7. Laboratory of Experimental Oncology, Dept. of Pathology, Erasmus MC, Rotterdam, The Netherlands.
8. Pharmaceutics Division, Dept. of Pharmaceutical Sciences, Faculty of Science, Utrecht University, Utrecht, The Netherlands.
Abstract
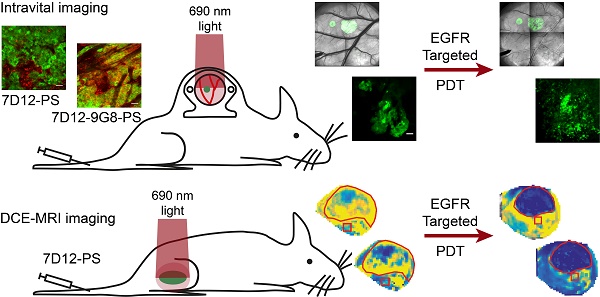
Targeted photodynamic therapy (PDT) has the potential to selectively damage tumor tissue and to increase tumor vessel permeability. Here we characterize the tissue biodistribution of two EGFR-targeted nanobody-photosensitizer conjugates (NB-PS), the monovalent 7D12-PS and the biparatopic 7D12-9G8-PS. In addition, we report on the local and acute phototoxic effects triggered by illumination of these NB-PS which have previously shown to lead to extensive tumor damage.
Methods: Intravital microscopy and the skin-fold chamber model, containing OSC-19-luc2-cGFP tumors, were used to investigate: a) the fluorescence kinetics and distribution, b) the vascular response and c) the induction of necrosis after illumination at 1 or 24 h post administration of 7D12-PS and 7D12-9G8-PS. In addition, dynamic contrast enhanced magnetic resonance imaging (DCE-MRI) of a solid tumor model was used to investigate the microvascular status 2 h after 7D12-PS mediated PDT.
Results: Image analysis showed significant tumor colocalization for both NB-PS which was higher for 7D12-9G8-PS. Intravital imaging showed clear tumor cell membrane localization 1 and 2 h after administration of 7D12-9G8-PS, and fluorescence in or close to endothelial cells in normal tissue for both NB-PS. PDT lead to vasoconstriction and leakage of tumor and normal tissue vessels in the skin-fold chamber model. DCE-MRI confirmed the reduction of tumor perfusion after 7D12-PS mediated PDT. PDT induced extensive tumor necrosis and moderate normal tissue damage, which was similar for both NB-PS conjugates. This was significantly reduced when illumination was performed at 24 h compared to 1 h after administration.
Discussion: Although differences were observed in distribution of the two NB-PS conjugates, both led to similar necrosis. Clearly, the response to PDT using NB-PS conjugates is the result of a complex mixture of tumor cell responses and vascular effects, which is likely to be necessary for a maximally effective treatment.
Keywords: Targeted, EGFR, Nanobody, Photosensitizer, Photodynamic therapy, Intravital microscopy.
 Global reach, higher impact
Global reach, higher impact