13.3
Impact Factor
Theranostics 2020; 10(6):2495-2508. doi:10.7150/thno.41225 This issue Cite
Research Paper
An intestinal model with a finger-like villus structure fabricated using a bioprinting process and collagen/SIS-based cell-laden bioink
Department of Biomechatronic Engineering College of Biotechnology and Bioengineering Sungkyunkwan University (SKKU), Suwon 16419, South Korea.
Abstract
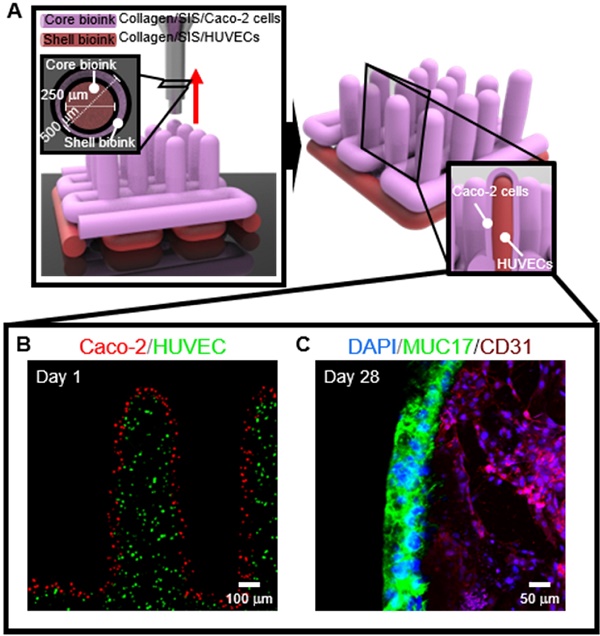
The surface of the small intestine has a finger-like microscale villus structure, which provides a large surface area to realize efficient digestion and absorption. However, the fabrication of a villus structure using a cell-laden bioink containing a decellularized small intestine submucosa, SIS, which can induce significant cellular activities, has not been attempted owing to the limited mechanical stiffness, which sustains the complex projective finger-like 3D structure. In this work, we developed a human intestinal villi model with an innovative bioprinting process using a collagen/SIS cell-laden bioink.
Methods: A Caco-2-laden microscale villus structure (geometry of the villus: height = 831.1 ± 36.2 μm and diameter = 190.9 ± 3.9 μm) using a bioink consisting of collagen type-I and SIS was generated using a vertically moving 3D bioprinting process. By manipulating various compositions of dECM and a crosslinking agent in the bioink and the processing factors (printing speed, printing time, and pneumatic pressure), the villus structure was achieved.
Results: The epithelial cell-laden collagen/SIS villi showed significant cell proliferation (1.2-fold) and demonstrated meaningful results for the various cellular activities, such as the expression of tight-junction proteins (ZO-1 and E-cadherin), ALP and ANPEP activities, MUC17 expression, and the permeability coefficient and the glucose uptake ability, compared with the pure 3D collagen villus structure.
Conclusion: In vitro cellular activities demonstrated that the proposed cell-laden collagen/dECM villus structure generates a more meaningful epithelium layer mimicking the intestinal structure, compared with the pure cell-laden collagen villus structure having a similar villus geometry. Based on the results, we believe that this dECM-based 3D villus model will be helpful in obtaining a more realistic physiological small-intestine model.
 Global reach, higher impact
Global reach, higher impact