Impact Factor
Theranostics 2020; 10(6):2522-2537. doi:10.7150/thno.39553 This issue Cite
Research Paper
Therapeutic ultrasound combined with microbubbles improves atherosclerotic plaque stability by selectively destroying the intraplaque neovasculature
1. Department of Cardiology, State Key Laboratory of Organ Failure Research, Nanfang Hospital, Southern Medical University, Guangzhou, China.
2. Guangzhou Regenerative Medicine and Health Guangdong Laboratory, 510005 Guangzhou, China.
3. Department of Cardiology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, China.
4. Department of Oncology, Nanfang Hospital, Southern Medical University, Guangzhou, China.
* Xinzhong Li and Shengcun Guo contributed equally to this work.
Abstract
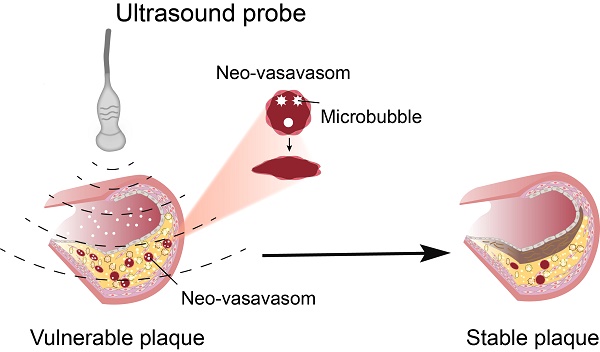
Objective: The current antiangiogenic therapy for atherosclerotic plaques was mainly achieved by the use of antiangiogenic drugs, but serious side effects have limited the clinical application. The present study investigated whether therapeutic ultrasound (TUS) treatment with appropriate pressure could selectively deplete the neovasculature in vulnerable plaques to improve its stability with no side effects on the body; the underlying mechanisms were also explored.
Methods and Results: A mouse model of advanced atherosclerosis was generated by maintaining apolipoprotein E-deficient (ApoE-/-) mice on a hypercholesterolemic diet (HCD). Plaque, skeletal muscle, mesentery and skin tissue from 114 atheroma-bearing mice were subjected to sham therapy, an ultrasound application combined with microbubbles at four different ultrasound pressures (1.0, 2.0, 3.0, 5.0 MPa), or ultrasound at 5.0 MPa alone. Microvessel density (MVD) was assessed by immunofluorescence and immunohistochemical methods. The plaque necrotic center/fiber cap (NC/FC) ratio and vulnerability index were calculated to evaluate plaque vulnerability. Twenty-four hours after TUS treatment at 3.0 MPa, the MVD in the plaque was substantially decreased by 84% (p < 0.05), while there was almost no change in MVD and neovessel density (NVD) in normal tissues, including skeletal muscle, mesentery and skin. Additionally, a marked reduction in the number of immature vessels was observed in the plaques (reduced by 90%, p < 0.05), whereas the number of mature vessels was not significantly decreased. Furthermore, TUS treatment at 3.0 MPa significantly improved plaque stability, as reflected by the NC/FC ratio and vulnerability index, which may be due to the selective destruction of intraplaque neovascularization by TUS treatment, thereby decreasing the extravasation of erythrocytes and leading to vascular inflammation alleviation and thin-cap fibroatheroma reduction.
Conclusions: TUS treatment at 3.0 MPa selectively depleted plaque neovessels and improved the stability of vulnerable plaques through a reduction in erythrocyte extravasation and inflammatory mediator influx, with no significant effect on normal tissue.
Keywords: therapeutic ultrasound, microbubbles, atherosclerosis, angiogenesis, plaque stability.
 Global reach, higher impact
Global reach, higher impact